If the wall in Fig. 4.29 is 4 m long, calculate the total force on the wall
Question:
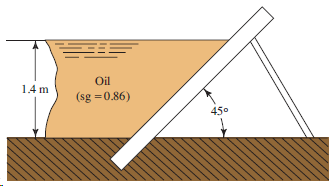
Transcribed Image Text:
Oil 14 m (sg =0.86) 450
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
F R 468 kN h p 23 h ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
If the cord attached to the vertical wall in Fig. 8.44 were horizontal (instead of at a angle), what would the tensions in the cords be? cord 1 45 30cord 2 1m 1.5 kg
-
Calculate the total force on the bottom of the closed tank shown in Fig. 4.23 if the air pressure is 52 kPa(gage). Air 0,50 m Oil (sg -0.85) 0.75 m Water 18 m 1.2 m
-
In Fig. E6.7 assume that there is no friction force on the 20.0-N block that sits on the tabletop. The pulley is light and frictionless. (a) Calculate the tension T in the light string that connects...
-
Builder Products, Incorporated, uses the weighted-average method in its process costing system. It manufactures a caulking compound that goes through three processing stages prior to completion....
-
Ryou Kato owns Kato Blankets. Ryou asks you to explain how he should treat the following reconciling items when reconciling the company's bank account. 1. Outstanding checks. 2. A deposit in transit....
-
J.D. Power and Associates calculates and publishes various statistics concerning car quality. The initial quality score measures the number of problems per new car sold. For 2012 model cars, Ford had...
-
a. What sampling units may be used when the audit objective is (1) proper executing and (2) proper recording of sales transactions? b. When is it preferable to use (a) shipping documents and (b)...
-
The CPA Partnership operated by Cook, Parks, and Argo is being liquidated. A balance sheet prepared at this stage in their liquidation process is presented below. The partners share profits and...
-
The invention relates to a community pass and method of use for the prevention of the spread of disease during a pandemic. People enter and exit the community at random and the recording and report...
-
Mandy Peters, the lead accountant of Ross Co., would like to buy a new general ledger software program. She couldnt do it because all funds were frozen for the rest of the fiscal period. Mandy called...
-
The wall shown in Fig. 4.28 is 20 ft long. (a) Calculate the total force on the wall due to water pressure and locate the center of pressure; (b) Calculate the moment due to this force at the base of...
-
Explain the difference between optimistic and pessimistic locking.
-
Do branding, packaging, and labeling really benefit consumers? Explain.
-
Although consumption of carbonated beverages has been decreasing, it is still an $81 billion industry in North America, which far exceeds alternatives such as water ($23 billion) and sports drinks...
-
Create chronological and functional rsums to highlight your key selling points.
-
Describe a firm that still uses a lot of television/print advertising. a. Is its marketing spending ineffective? b. Why do you think the firm still allocates resource to this area? c. How would you...
-
What is casting? How it differs from other primary shaping processes?
-
Evaluate the convolution \(t * u(t-a)\).
-
Determine whether A is diagonalizable and, if so, find an invertible matrix P and a diagonal matrix D such that P -1 AP = D. -3 4 A -1
-
Consider the discrete group G of order 8 that has the following Cayley diagram e If we have the sequence of operations: fcagec, which of the options represents the reduction of the sequence to a...
-
From the following data at 298.15 K as well as data in Table 4.1 (Appendix B, Data Tables), calculate the standard enthalpy of formation of H 2 S(g) and of FeS 2 (s): AR(kJ mol) Fe(s) + 2H2S(g) ...
-
Which of Ne or Ar has the larger van der Waals parameter b? Explain your reasoning.
-
You have calculated the pressure exerted by ethane using the ideal gas law and the RedlichKwong equations of state. How do you decide if the repulsive or attractive part of the molecular potential...
-
Consider a 15%, 20 year bond that pays interest annually, and its current price is $850. What is the promised yield to maturity?
-
You are a dual-income, no-kids family. You and your spouse havethe following debts (total): mortgage, $212,000; auto loan,$22,000; credit card balance, $16,000; other debts, $22,000.Further, you...
-
Can you please provide a feedback to this post? 1. What is the managerial view on business? What are the stakeholders view on business? How do they differ? The management viewpoint on business...

Study smarter with the SolutionInn App


