Repeat Problem 4.47 using Fig. 4.47, except that there is now 7.50 kPa air pressure above the
Question:
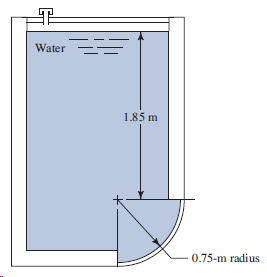
Transcribed Image Text:
Water 1.85 m 0.75-m radius
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
h 1e h 1 h a 185 0765 2615m h ce h 1e s2 2615 ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Repeat Problem 4.48 using Fig. 4.48, except that there is now 4.65 kPa air pressure above the fluid. Ammonia sg = 0.826 0,62 m 1.25 m
-
For the system described in Problem 11.47, and using the tube size found in that problem, compute the expected volume flow rate through the tube if the pressure above the fluid in the large tank at A...
-
In Problem 7.8 (Fig. 7.17), if the left-hand tank is also sealed and air pressure above the water is 68 kPa, calculate the pump power. In Problem A pump is being used to transfer water from an open...
-
QUESTION 5 Directions: Passage B is the story of a childs first Halloweenparade. Read the passage carefully. Answer the questions thatfollow. Passage B I am six years old. It is Halloween. The school...
-
Howsham Company, Ltd. reports the following for the month of June. Instructions (a) Calculate the cost of the ending inventory and the cost of goods sold for (1) FIFO and (2) moving-average cost,...
-
Is the number of calories in a beer related to the number of carbohydrates and/or the percentage of alcohol in the beer? Data concerning 150 of the best-selling domestic beers in the United States...
-
What role does a data control group play in an EDP system?
-
King Corporation began operations in January 2011. The charter authorized the following capital stock: Preferred stock: 10 percent, $10 par, authorized 40,000 shares Common stock : $5 par, authorized...
-
Describe the process termination sequence in an operating system. How does the OS ensure that resources are properly released, and what mechanisms are used to manage orphaned processes or zombie...
-
John and Martha Holloway are married filing jointly. They are 35 and 31 years old, respectively. Their address is 10010 Dove Street, Atlanta, GA 30294. Additional information about Mr. and Mrs....
-
Use Fig. 4.54. The surface is 60 in long. 48 in 36 in Alcohol sg = 0.79
-
The tank in Fig. 4.55 has a view port in the inclined side. Compute the magnitude of the resultant force on the panel. Show the resultant force on the door clearly and dimension its location. 20 in...
-
How would your answer in question 6 change if the alternatives read with a relatively high or with a relatively low marginal leakage rate? Explain.
-
A \(27-\mathrm{kg}\) child stands in the center of a trampoline. (a) If the trampoline center is \(0.32 \mathrm{~m}\) lower than before they got on, what is the spring constant of the trampoline? (b)...
-
Describe the various ways in which the courts become involved with separation agreements.
-
An 80-kg man rides in an elevator from the ground to an upper floor. The elevator goes from rest to \(10 \mathrm{~m} / \mathrm{s}\) in 5 seconds, and then continues at the same constant speed for 10...
-
Describe how the courts historically viewed separation agreements and why.
-
You throw a \(100-\mathrm{g}\) ball upward with a speed of \(19.8 \mathrm{~m} / \mathrm{s}\). How much work does the force of gravity do on the ball during its trip to its maximum height?
-
Compute the determinants using cofactor expansion along the first row and along the first column. -2 3 3 -1
-
An annual report of The Campbell Soup Company reported on its income statement $2.4 million as equity in earnings of affiliates. Journalize the entry that Campbell would have made to record this...
-
Propose a plausible mechanism for the reaction below: [H2SO4] N- - . -N.
-
As shown above, methenamine is hydrolyzed in aqueous acid to produce formaldehyde and ammonia. Draw a mechanism showing formation of one molecule of formaldehyde (the remaining five molecules of...
-
Predict the major product for each reaction below: (a) (b) 1) [H'), HS 2) Raney Ni SH 1) [H*), HS' `H. 2) Raney Ni SH
-
Provide an analysis of the benefits and challenges of having a growth mindset as opposed to having a fixed mindset. Examine the benefits of each type of mindset, as well as the challenges that each...
-
Discuss and explain what is Performance Management (P M)? How does P M fit into corporate strategy? What's in it for me? How does it work? What are my responsibilities? How does PM relate to other...
-
A conducting spherical shell has inner radius 2 / 3 R and outer radius R . ( a ) Suppose we put a charge Q onthe shell ( a conductor ) so that all of the charge goes to the outer radius. Find the...

Study smarter with the SolutionInn App


