Repeat Problem 4.48 using Fig. 4.48, except that there is now 4.65 kPa air pressure above the
Question:
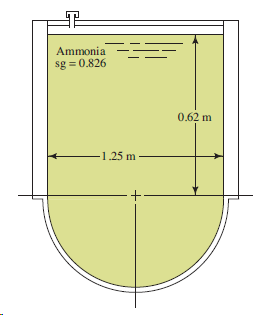
Transcribed Image Text:
Ammonia sg = 0.826 0,62 m 1.25 m
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
h 1e h 1 h a 062 m 0574 m 1194 m ...View the full answer

Answered By

Anjali Arora
Having the experience of 16 years in providing the best solutions with a proven track record of technical contribution and appreciated for leadership in enhancing team productivity, deliverable quality, and customer satisfaction. Expertise in providing the solution in Computer Science, Management, Accounting, English, Statistics, and Maths.
Also, do website designing and Programming.
Having 7 yrs of Project Management experience.
100% satisfactory answers.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Repeat Problem 4.47 using Fig. 4.47, except that there is now 7.50 kPa air pressure above the fluid. Water 1.85 m 0.75-m radius
-
For the system described in Problem 11.47, and using the tube size found in that problem, compute the expected volume flow rate through the tube if the pressure above the fluid in the large tank at A...
-
In Problem 7.8 (Fig. 7.17), if the left-hand tank is also sealed and air pressure above the water is 68 kPa, calculate the pump power. In Problem A pump is being used to transfer water from an open...
-
Which of these is located farthest south? Crete O Sardinia Sicily Corsica
-
The cost of goods sold computations for Gouda Company NV and Edam Company NV are shown below. Instructions (a) Compute inventory turnover and days in inventory for each company. (b) Which company...
-
Hydro Systems Engineering Associates, Inc. provides consulting services to city water authorities. The consulting firms contribution-margin ratio is 20 percent, and its annual fixed expenses are...
-
Baltimore Realty, Inc., had the following transactions for the month of June 2010. The following T-accounts have been set up, for Baltimore Realty, Inc., with their beginning balances as of June 1,...
-
The financial statements of Tootsie Roll Industries are presented in Appendix A. Instructions Answer the following questions. (a) What was the amount of net cash provided by operating activities for...
-
The Snow Lover Company manufactures a single product, the ski board. The budgeted selling price is $30 per unit. The annual budgeted fixed manufacturing overhead is $3,600,000. Snow Lover has zero...
-
Consider two ideas (v,c), where idea 1 is (5,25), and idea 2 is (4,15). Suppose that, at most, one idea may be selected. We are using the efficiency of the ideas to determine which idea should be...
-
Use Fig. 4.54. The surface is 60 in long. 48 in 36 in Alcohol sg = 0.79
-
The tank in Fig. 4.55 has a view port in the inclined side. Compute the magnitude of the resultant force on the panel. Show the resultant force on the door clearly and dimension its location. 20 in...
-
Suppose that the random variable X has mean and variance 2. Show that the third central moment of X can be expressed as E(X3) 32 3.
-
Briefly describe how this phase links in with the Foundations and Enablement phases and with the BPM House.
-
What is the relative importance of family sponsorship (of permanent residency applications) in the United States as compared with Australia and Canada? What factor do Australia and Canada emphasize...
-
Explain the relationship between sustainability and poverty.
-
Outline three of the Sustainable Development Goals.
-
Consider the situation in Merton's jump-diffusion model where the underlying asset is a non-dividend-paying stock. The average frequency of jumps is one per year. The average percentage jump size is...
-
Describe a recycled product that can be produced from each type of food service- generated solid waste.
-
1. Following are information about Alhadaf Co. Cost incurred Inventory Purchases Sales Adverting expense Salary Expense Depreciation Beginning Inventory Ending Inventory Amount 118,000 350.000 90,000...
-
Draw the structure of the cyclic compound that is produced when acetone is treated with 1,3 propanedithiol in the presence of an acid catalyst. SH 1,3-propanedithiol HS Acetone
-
Predict the major product for each of the following reactions: (a) (b) (c) (d) 1) LAH 2) H20 H NABH4,
-
When 2 moles of benzaldehyde are treated with sodium hydroxide, a reaction occurs in which 1 mole of benzaldehyde is oxidized (giving benzoic acid) while the other mole of benzaldehyde is reduced...
-
The Dougherty Furniture Company manufactures tables. In March, the production department had budgeted allocation base (i.e. cost driver) of 4,000 machine-hours. The budgeted manufacturing overheads...
-
Consider the following pseudocode for calculating ab, where a and b are positive integers. 19 FastPower Input: positive integers a and b. Output: ab. return a if b=1 then else c:=b.b...
-
Consider one disk with 2000 cylinders, numbered 0 to 1 999. Assume the current position of head is at cylinder 500. The request queue is given as follows: 103, 80, 1400, 813, 1714, 748, 1500, 1100...

Study smarter with the SolutionInn App


