Show that the reaction Glucose 2 Glyceraldehyde-3-phosphate is slightly endergonic (G' = 2.2 kJ mol -1
Question:
Show that the reaction Glucose → 2 Glyceraldehyde-3-phosphate is slightly endergonic (ΔG°' = 2.2 kJ mol-1 = 0.53 kcal mol-1); that is, it is not too far from equilibrium. Use the data in Table 17.1.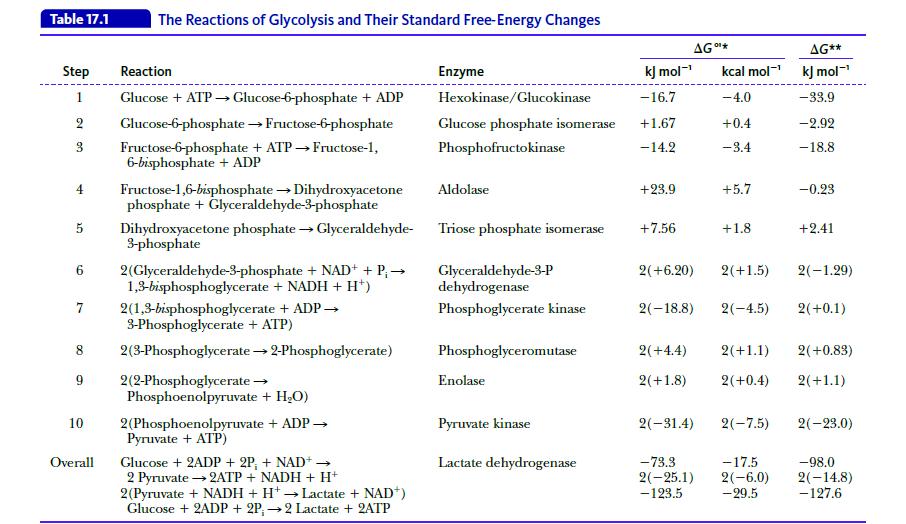
Transcribed Image Text:
Table 17.1 Step Reaction 1 Glucose + ATP → Glucose-6-phosphate + ADP 2 Glucose-6-phosphate → Fructose-6-phosphate 3 Fructose-6-phosphate + ATP→Fructose-1, 6-bisphosphate + ADP 4 Fructose-1,6-bisphosphate → Dihydroxyacetone phosphate + Glyceraldehyde-3-phosphate 5 Dihydroxyacetone phosphate → Glyceraldehyde- 3-phosphate 6 2(Glyceraldehyde-3-phosphate + NAD+ + P₁ → 1,3-bisphosphoglycerate + NADH + H¹) 7 The Reactions of Glycolysis and Their Standard Free-Energy Changes 8 9 10 Overall 2(1,3-bisphosphoglycerate + ADP → 3-Phosphoglycerate + ATP) 2(3-Phosphoglycerate → 2-Phosphoglycerate) 2(2-Phosphoglycerate → Phosphoenolpyruvate + H₂O) 2(Phosphoenolpyruvate + ADP → Pyruvate + ATP) Glucose + 2ADP + 2P, + NAD+ → 2 Pyruvate → 2ATP + NADH + H+ - 2(Pyruvate + NADH + H+ → Lactate + NAD+) Glucose + 2ADP + 2P;→2 Lactate + 2ATP Enzyme kJ mol-¹ Hexokinase/Glucokinase -16.7 Glucose phosphate isomerase +1.67 Phosphofructokinase -14.2 Aldolase Triose phosphate isomerase Glyceraldehyde-3-P dehydrogenase Phosphoglycerate kinase Phosphoglyceromutase Enolase Pyruvate kinase Lactate dehydrogenase +23.9 AG *** +7.56 kcal mol-¹ -4.0 +0.4 -3.4 2(-18.8) +5.7 +1.8 2(+6.20) 2(+1.5) 2(-4.5) 2(+4.4) 2(+1.8) 2(+0.4) 2(-31.4) 2(-7.5) AG** kJ mol-¹ -33.9 -2.92 -18.8 -73.3 -17.5 2(−25.1) 2(-6.0) -123.5 - 29.5 -0.23 +2.41 2(+1.1) 2(+0.83) 2(+1.1) 2(-1.29) 2(+0.1) 2(-23.0) -98.0 2(-14.8) -127.6
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Add the G mol 1 values f...View the full answer

Answered By

Hassan Ali
I am an electrical engineer with Master in Management (Engineering). I have been teaching for more than 10years and still helping a a lot of students online and in person. In addition to that, I not only have theoretical experience but also have practical experience by working on different managerial positions in different companies. Now I am running my own company successfully which I launched in 2019. I can provide complete guidance in the following fields. System engineering management, research and lab reports, power transmission, utilisation and distribution, generators and motors, organizational behaviour, essay writing, general management, digital system design, control system, business and leadership.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Biochemistry
ISBN: 9781305961135
9th Edition
Authors: Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
For the reaction N 2 (g) + 3 H 2 (g) 2 NH 3 (g), the equilibrium constant is K p = 36.5 at 400 K. Two separate equilibrium mixtures have the following compositions at 400 K and a total pressure of...
-
The first nuclear reaction ever observed (in 1919 by Ernest Rutherford) was a + 147N p+ X. (a) Show that the reaction product "X" must be 178O. (b) For this reaction to take place, the particle...
-
Why program planning is important in public health leadership?
-
The Cutting Department of Hong Manufacturing has the following production and cost data for July. Instructions(a) Determine the equivalent units of production for (1) Materials and (2) Conversion...
-
Using the Internet and a Web browser, visit a search engine such as Google or Yahoo! and type marketing research. From the thousands of options you are offered, pick a website that you find...
-
What are the two possible translation processes that may be necessary to translate a foreign operations financial statements?
-
Congratulations! You have won a state lotto. The state lottery offers you the following (after-tax) payout options: Option #1: $15,000,000 after five years. Option #2: $2,150,000 per year for the...
-
3. You purchased a 3 year coupon bond one year ago. Its par value is $1,000 and coupon rate is 6%, paid annually. At the time you purchased the bond, its yield to maturity was 6.5%. Suppose you sell...
-
What are some of the main differences between the cell walls of plants and those of bacteria?
-
A friend asks you why some parents at her childs school want a choice of beverages served at lunch, rather than milk alone. What do you tell your friend?
-
Consider an ideal gas turbine cycle with two stages of compression and two stages of expansion. The pressure ratio across each stage of the compressor and the turbine is 2. Air (use the IG model)...
-
How/why are banks and non-bank loan-making companies using risk transfer securities (like the securitization of yacht loans by Credit Swiss)? Who will bear the final loss if there are defaults or...
-
If a company decides to implement a Multinational Netting: Why does it need to implement organizational controls (2 elements point-form)?
-
How is information used for decision making? How managers approach decisions? Please explain "decision conditions" B) Please explain, the decision making process. (Steps in decision making) Do the...
-
Define a problem, problem identification, problem solving, and decision making and discuss types of decisions and decision-making conditions. Discuss the nature, and process of decision making
-
When selecting a banking partner the company must analyze both the Counterparty risk and Operational risk; please provide an example for both risks
-
Williams Manufacturing uses scrap metal to produce various tools, such as drill bits, hammer heads, saw blades, and nails. The CEO has asked you to analyze the saw blades division to determine asset...
-
You are maintaining a subsidiary ledger account for Police-Training Expenditures for 2013. The following columns are used: Inventory purchases are initially recorded as expenditures. Record the...
-
A small DNA molecule was cleaved with several dierent restriction nucleases, and the size of each fragment was determined by gel electrophoresis. The following data were obtained. Enzyme Fragment...
-
Te average human chromosome contains about 1 108 bp of DNA. (a) If each base pair has a mass of about 660 daltons and there are about 2 g of protein (histones plus nonhistones) per gram of DNA, how...
-
Forming nucleosomes and wrapping them into a 30-nm ber provide part of the compaction of DNA in chromatin. If the ber contains about six nucleosomes per 10 nm of length, what is the approximate...
-
PRP obtained a $ 1 0 million convertible mortgage to finance the aquisition of a 1 2 . 5 million office building. The mortgage features a fixed interest rate of 9 % over 2 5 years, with payments due...
-
Mike was worried about the backlog of overdue Vortex orders at the Rosemount Measurement plant. Some customers were very concerned, and some had considered using other suppliers, because the plant...
-
1. HR strategy is the most important functional strategy, without synergy with the business strategy, the vision and long-term objectives cannot be implemented? ( 2. Ratio analysis, is an effective...

Study smarter with the SolutionInn App


