Add labels to the figure that follows, which illustrates the subatomic particles associated with a carbon atom.
Question:
Add labels to the figure that follows, which illustrates the subatomic particles associated with a carbon atom.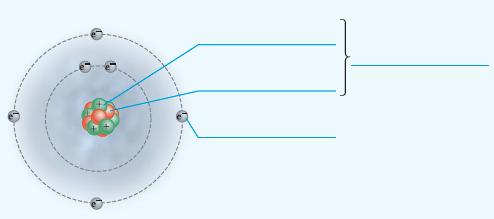
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Proton ...View the full answer

Answered By

Sinmon Warui Kamau
After moving up and down looking for a job, a friend introduced me to freelance writing. I started with content writing and later navigated to academic writing. I love writing because apart from making a living out of it, it is also a method of learning and helping others to learn.
5.00+
40+ Reviews
45+ Question Solved
Related Book For 

Biology Science For Life With Physiology
ISBN: 9780134555430
6th Edition
Authors: Colleen Belk, Virginia Maier
Question Posted:
Students also viewed these Sciences questions
-
Add labels to the figure that follows, which illustrates how Mycobacterium tuberculosis evolves when it is exposed to an antibiotic. Single drug therapy The initial M. tuberculosis population...
-
Add labels to the figure that follows, which illustrates a transfer RNA molecule. tRNA AAA UUU
-
Add labels to the figure that follows, which illustrates female internal reproductive organs.
-
In a test on 2000 electric bulbs, it was found that the life of a particular make was normally distributed with an average life of 2040 hos & S.D of bohos Estimate the ne of likely to burn for it...
-
Consider the fuel element of Example 5.9, which operates at a uniform volumetric generation rate of q) = 10 7 W/m3, until the generation rate suddenly changes to q2 = 2 x 107 W/m3. Use the...
-
Do you think religion can affect work behaviors?
-
The magnitude of the osmotic pressure depends on the (a) Temperature (b) Gibbs' free energy (c) Nature of the semi-permeable membrane (d) Entropy.
-
Hill Company uses budgets in controlling costs. The August 2012 budget report for the companys Assembling Department is as follows. The monthly budget amounts in the report were based on an expected...
-
On January 1, a company agrees to pay $28,000 in four years. If the annual interest rate is 5%, determine how much cash the company can borrow with this agreement. (Use appropriate factor(s) from...
-
Water ___________. A. Is a good solute; B. Facilitates chemical reactions; C. Serves as an enzyme; D. Makes strong covalent bonds with other molecules; E. Consists of two oxygen and one hydrogen atoms
-
List the structural features in a prokaryotic cell.
-
You are conducting an external audit of the companys financial statements and are working at the clients office two weeks after the year end. a. Design a test with two audit procedures, in addition...
-
Future Shop has a deal on new computer tables. If you buy the table, they ask you to pay $160 today. After that, they ask you to make 12 monthly payments of $180 starting in 1 month. Suppose you...
-
Capital Budgeting Case / Problem Parameters: A capital project and budgeting decision is being considered that would involve an expansion, along with a replacement of some old equipment. The project...
-
Write a note on following languages, their standards, compilers and IDEs which support them. C# C++ Java Fortran Pascal Ruby PHP COBOL Python [Marks 10]
-
Apartment occupancy rates in a coastal town are thought to be influenced by the number of tourists staying in the area. Over the past 10 years, the following data has been collected: Year Number of...
-
1- What may be the steps of marketing research to decide where to open a new branch of a major bank? A detail analysis. 2-Which one of the five major marketing concepts would you use when you market...
-
Ramona owns 20% of the stock of Miller, Inc. Miller reports the following items for the current year: Sales $3,400,000 Gain on sale of stock held for 2 years 250,000 Cost of goods sold 1,800,000...
-
2. In the circuit given in Figure 2, i,(t) = 5.67cos(5t)A and v (t) = 70.71 cos(5t 60) V a) Find the equivalent load impedance. State whether the load is inductive or capacitive. b) Calculate the...
-
Use resonance structures to help you identify all sites of low electron density (δ+) in the following compound:
-
Liquid N 2 has a density of 875.4 kg m 3 at its normal boiling point. What volume does a balloon occupy at 298 K and a pressure of 1.00 atm if 3.10 10 3 L of liquid N 2 is injected into it? Assume...
-
Calculate the volume of all gases evolved by the complete oxidation of 0.375 g of the amino acid alanine NH 2 CHCH 3 COOH if the products are liquid water, nitrogen gas, and carbon dioxide gas, the...
-
The following transactions occurred at the Daisy King Ice Cream Company. 1. Started business by issuing 10,000 shares of common stock for $35,000. 2. Leased a building for three years at $650 per...
-
Elaborate on future opportunities, what you have learned from this exercise, and how you will use the experience gained in this exercise to improve your revenue management critical-thinking process
-
Angela Lopez owns and manages a consulting firm called Metrix, which began operations on December 1. On December 31, Metrix shows the following selected accounts and amounts for the month of...

Study smarter with the SolutionInn App


