Describe a series of steps in which you use the cylinder of Figure 17.13 to implement the
Question:
Describe a series of steps in which you use the cylinder of Figure 17.13 to implement the ideal-gas process shown in Figure Q 17.10. Then show the process as a first-law bar chart.
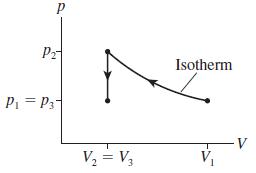
Figure Q 17.10
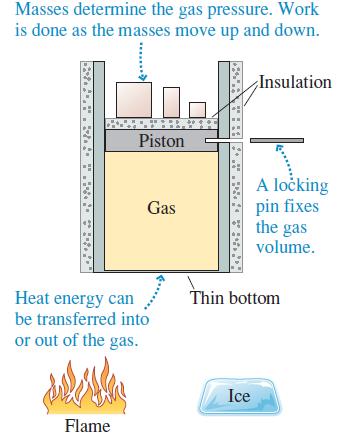
Figure 17.13
Transcribed Image Text:
Pz Isotherm Pi = P3- V, = V3 V V,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
For the isothermal process T 0 K This means the first law of thermodynamics can only be satisfie...View the full answer

Answered By

ALBANUS MUTUKU
If you are looking for exceptional academic and non-academic work feel free to consider my expertise and you will not regret. I have enough experience working in the freelancing industry hence the unmistakable quality service delivery
4.70+
178+ Reviews
335+ Question Solved
Related Book For 

Physics For Scientists And Engineers A Strategic Approach With Modern Physics
ISBN: 9780321740908
3rd Edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Mathematics questions
-
Identify three situations in which you use self-service delivery. For each situation, what is your motivation for using self-service delivery, rather than having service personnel do it for you?
-
Consider a borrow-and-invest strategy in which you use $1 million of your own money and borrow another $1 million (at the t-bill rate) to invest $2 million in a market index fund. If the risk free...
-
In a cylinder filled up with ideal gas and closed from both ends there is a piston of mass m and cross-sectional area S (Fig. 4.21). In equilibrium the piston divides the cylinder into two equal...
-
Visit any social media site with news stories that includes a photograph/snapshot of story (mandatory) . Choose ONE news story. Use the questions below to help you determine validity of that ONE news...
-
An ideal vapor-compression refrigeration cycle that uses refrigerant-134a as its working fluid maintains a condenser at 800 kPa and the evaporator at - 12oC. Determine this system's COP and the...
-
The amounts of the assets and liabilities of Viva Travel Service as of September 30, 20Y6, the end of the current year, and its revenue and expenses for the year are listed below. The retained...
-
In a vapour compression refrigeration system, the lowest temperature during the cycle occurs after (a) compression (b) condensation (c) expansion (d) evaporation
-
A parent company paid $500,000 for a 100% interest in a subsidiary. At the end of the first year, the subsidiary reported net income of $40,000 and paid $5,000 in dividends. The price paid reflected...
-
Describe the behaviours that individual team members can engage in that positively impact overall team effectiveness.
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Describe a series of steps in which you use the cylinder of Figure 17.13 to implement the ideal-gas process shown in Figure Q 17.9. Then show the process as a first-law bar chart. Figure Q 17.9...
-
How much heat energy must be added to a 6.0 cm 6.0 cm 6.0 cm block of aluminum to raise its temperature from - 50C to 50C?
-
Considering issues raised in this chapter, under what circumstances might the HSMC not be appropriate and why? Could some of these issues be overcome so as to be able to use this phased approach?
-
You are an entrepreneur starting a biotechnology firm. If your research is successful, the technology can be sold for $30 million. If your research is unsuccessful, it will be worth nothing. To fund...
-
What is your opinion of organizations using bona fide occupational qualifications (BFOQs) to limit who they will consider for a job?
-
In your opinion, is most discrimination in the United States unintentional (disparate impact), or is most discrimination intentional (disparate treatment)? Why do you think so?
-
Find two outside examples of how companies created new product lines as a result of identifying market opportunities or problems.
-
List three types of visuals and their purposes. How do visuals enhance marketing research reports?
-
Review Conceptual Example 11 before starting to solve this problem. The number of unstable nuclei remaining after a time t = 5.00 yr is N, and the number present initially is N 0 . Find the ratio N/N...
-
How will relating product contribution margin s to the amount of the constrained resource they consume help a company maximize its profits?
-
The cable cars in San Francisco are pulled along their tracks by an underground steel cable that moves along at 9.5 mph. The cable is driven by large motors at a central power station and extends,...
-
A 2.0 kg rope hangs from the ceiling. What is the tension at the midpoint of the rope?
-
A 2.0 kg rope hangs from the ceiling. What is the tension at the midpoint of the rope?
-
Korda International Inc. recently issued new securities (common shares and bonds) to finance a new project with a cost $16 million. The equity issued had a flotation cost of 8%, while the debt issued...
-
Khairul bought a television set with cash price of Rm6000. He paid a 10% down payment and the balance was settled by making a 24 monthly payments . If the interest was 8% per annum on the original...
-
Suppose a company produces a perpetual cash flow of $25 million per year and is expected to continue doing so in the infinite future. The company's capital structure currently consists entirely of...

Study smarter with the SolutionInn App


