Prove that the normalization constant of the 2p radial wave function of the hydrogen atom is (24a
Question:
Prove that the normalization constant of the 2p radial wave function of the hydrogen atom is (24πaB3)–1/2, as shown in Equations 41.7.
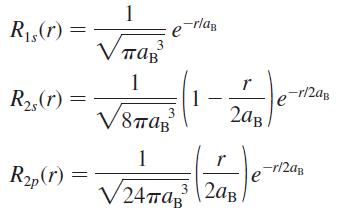
Transcribed Image Text:
1 R1,(r) = -rlag 1 ap -r/2ag R2,(r) 1 3 2ag 1 r R„(r) -r/2ag e V24ma, |2aB. 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
Solve From Equation 417 the 2p radia...View the full answer

Answered By

Khurram shahzad
I am an experienced tutor and have more than 7 years’ experience in the field of tutoring. My areas of expertise are Technology, statistics tasks I also tutor in Social Sciences, Humanities, Marketing, Project Management, Geology, Earth Sciences, Life Sciences, Computer Sciences, Physics, Psychology, Law Engineering, Media Studies, IR and many others.
I have been writing blogs, Tech news article, and listicles for American and UK based websites.
4.90+
5+ Reviews
17+ Question Solved
Related Book For 

Physics For Scientists And Engineers A Strategic Approach With Modern Physics
ISBN: 9780321740908
3rd Edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Mathematics questions
-
Prove that the normalization constant of the 1s radial wave function of the hydrogen atom is (a B 3 ) 1/2 , as given in Equations 41.7. A useful definite integral is
-
Prove that the normalization factor given by (12.2.12) ensures that (12.2.19) issatisfied. M-1 Ew?(n) (12.2.12) W(f)df = 1 J-1/2 (12.2.19)
-
The wave function for a hydrogen atom in the 2s state is(a) Verify that this function is normalized.(b) In the Bohr model, the distance between the electron and the nucleus in the n = 2 state is...
-
Contact local employers and ask for copies of their employee handbooks. If none are available, research parent companies of local employers online to see if their employee handbooks are available....
-
Determine the maximum amount of load, in kg, the balloon described in Prob. 1-102 can carry.
-
Repeat Problem 19 in Chapter 1, using Laplace transforms. Assume zero initial conditions. Data From Problem 19 in Chapter 1: Repeat Problem 18 using the network shown in Figure P1.7. Assume R=1;...
-
The table lists the number of wireless devices per household in a small town in the United States. (a) Construct a probability distribution. (b) Graph the probability distribution using a histogram...
-
At January 1, 2016, Hilly Mountain Flagpoles had Accounts Receivable of $31,000, and Allowance for Bad Debts had a credit balance of $3,000. During the year, Hilly Mountain Flagpoles recorded the...
-
Please discuss the role of an administrative healthcare professional and the skills and characteristics needed to perform this role successfully. Your response must be at least 10 well-written,...
-
These will be elaborated in the following sections. To illustrate better the computations of fmancial ratios and its interpretation, the financial statements if ABC Bhd is provide in TABLE 1.1....
-
Prove that the radial probability density peaks at r = a B for the 1s state of hydrogen.
-
For an electron in the 1s state of hydrogen, what is the probability of being in a spherical shell of thickness 0.010a B at distance (a) 1/2 a B (b) a B (c) 2a B from the proton?
-
(a) Determine the amount at which Fast should report each of the following on its balance sheet at December 31, Year 2, using (1) IFRS and (2) ASPE. Ignore the possibility of any additional...
-
a. What is the difference between liabilities and equity? b. What makes a liability a current liability? c. Provide some examples of current liabilities. d. What is the difference between long-term...
-
From a global economy perspective, what are the ethical issues that need to be considered to account for the possibility of conflicting issues and influences?
-
What traits do dominant people possess? Is there a best place for salespeople on the dominance continuum?
-
Bright Horizons Skilled Nursing Facility, an investor-owned company, constructed a new building to replace its outdated facility. The new building was completed on January 1, 2020, and Bright...
-
Although trust is an essential element of every sale, the meaning of trust changes with the type of sale. What are the three main types of sale, and how does the nature of trust change within the...
-
The steering wheel of a car has a radius of 0.19 m, and the steering wheel of a truck has a radius of 0.25 m. The same force is applied in the same direction to each steering wheel. What is the ratio...
-
Write out the formula for the total costs of carrying and ordering inventory, and then use the formula to derive the EOQ model. Andria Mullins, financial manager of Webster Electronics, has been...
-
The nucleus of the polonium isotope 214 Po (mass 214 u) is radioactive and decays by emitting an alpha particle (a helium nucleus with mass 4 u). Laboratory experiments measure the speed of the alpha...
-
A neutron is an electrically neutral subatomic particle with a mass just slightly greater than that of a proton. A free neutron is radioactive and decays after a few minutes into other subatomic...
-
A neutron is an electrically neutral subatomic particle with a mass just slightly greater than that of a proton. A free neutron is radioactive and decays after a few minutes into other subatomic...
-
A farmer used to sell a carton of eggs for $2.50 each. At this price, the farmer used to sell an average of 2,500 cartons of eggs per month. When the farmer raised the price to $3.20, sales dropped...
-
You pay $32,000 today (t=0) and obtain (purchase) a 9 percent annuity (r=0.09) with 24 annual payments (you will receive 24 payments). The first payment on the annuity is made in two years (t = 2)....
-
The bank loan schedule for financing the new office requires $1,600 payment at the beginning of each month for 2 years, starting in 18 months later. However, interest on the bank loan still charges...

Study smarter with the SolutionInn App


