The wave function for a hydrogen atom in the 2s state is(a) Verify that this function is
Question:
The wave function for a hydrogen atom in the 2s state is(a) Verify that this function is normalized.(b) In the Bohr model, the distance between the electron and the nucleus in the n = 2 state is exactly 4a. Calculate the probability that an electron in the 2s state will be found at a distance less than 4a from thenucleus.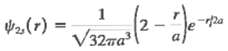
Transcribed Image Text:
1 √3²70²/(2²-²7) e-72² -pa
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 93% (15 reviews)
a Since the given r is real r lyf r The probability density wil...View the full answer

Answered By

WAHIDUL HAQUE
hello,
I'm a professional academic solution provider working as a freelance academic solution provider since 7 years. I have completed numerous projects. Help lots of students to get good marks in their exams and quizzes. I can provide any type of academic help to your homework, classwork etc, if you are a student of Accounting, Finance, Economics, Statistics. I believe in satisfying client by my work quality, rather than making one-time profit. I charge reasonable so that we make good long term relationship. why will you choose me? i am an extremely passionate, boldly honest, ethically driven and pro-active contractor that holds each of my clients in high regards throughout all my business relations. in addition, I'll always make sure that I'm giving my 100% better in every work that will be entrusted to me to be able to produce an outcome that will meet my client's standards. so if you are a student that is now reading my profile and considering me for your academic help. please feel free to look through my working history, feedback and contact me if you see or read something that interests you. I appreciate your time and consideration.
regards
4.90+
233+ Reviews
368+ Question Solved
Related Book For 

Physics
ISBN: 978-0077339685
2nd edition
Authors: Alan Giambattista, Betty Richardson, Robert Richardson
Question Posted:
Students also viewed these Modern Physics questions
-
The wave function for a traveling wave on a taut string is (in SI units) y(x, t) = (0.350 m) sin (10t 3x + /4) (a) What are the speed and direction of travel of the wave? (b) What is the vertical...
-
The wave function for a standing wave is given in Equation 18.3 as y = 2A sin kx cos wt. (a) Rewrite this wave function in terms of the wavelength ' and the wave speed v of the wave. (b) Write the...
-
The wave function for a wave on a taut string is y(x, t) = (0.350 m) s in (10t - 3x + /4) where x is in meters and t in seconds. (a) What is the average rate at which energy is transmitted along the...
-
Write the surface x 2 + y 2 z 2 = 2 (x + y) as an equation r = (, z) in cylindrical coordinates.
-
The following boxplot shows the residential fresh water use for all counties in Maryland, Louisiana, and Ohio in gallons per person per day. For example, in the most water frugal county in Maryland,...
-
Four charges are located at the corners of a square, as illustrated in Fig. 15.27. What are the magnitude and the direction of the force (a) On charge q2 and (b) On charge q4? q1 =-10pC 0.10 m 0.10 m...
-
A safety PLC has an Ethernet connection port used to send information to a data server in the system. Can this port be used without a cybersecurity risk?
-
Ashley Limited, which follows IFRS, chooses to classify interest and dividends received as well as interest paid as operating activities and dividends paid as financing activities. Ashley had the...
-
A 79.0 kg person is riding a bike on a level horizontal street. Together, the bike and person have a mass of 90.1 kg and a speed of 3.00 m/s. The person jumps off the bike, and just after the jump...
-
1. Let us complicate the story of Jokx and Beljeau enormously by sup- posing that both Jokx and Beljeau must decide whether to commission market surveys that will reveal the size of the market. For...
-
The wave function for a hydrogen atom in the 2s state is(a) Verify that this function is normalized.(b) In the Bohr model, the distance between the electron and the nucleus in the n = 2 state is...
-
(a) For an excited state of hydrogen, show that the smallest angle that the orbital angular momentum vector L can have with the z-axis is(b) What is the corresponding expression for (?L)max the...
-
Often research and development costs provide companies with benefits that last a number of years. (For example, these costs can lead to the development of a patent that will increase the companys...
-
An economist for the Brookings Institution argued that a price on carbon would minimize the cost of steering economic activity away from the greenhouse gas emissions that threaten the climate. a. In...
-
Corrs Wholesalers Co. sells industrial equipment for a standard 3-year note receivable. Revenue is recognized at time of sale. Each note is secured by a lien on the equipment and has a face amount...
-
On July 1, 2015, Wallace Company, a calendar-year company, sold special-order merchandise on credit and received in return an interest-bearing note receivable from the customer. Wallace Company will...
-
Select indicators from Table 10-3 that would be useful for the titrations in Figures 10-1 and 10-2 and the pK a = 8 curve in Figure 10-3. Select a different indicator for each titration and state...
-
William Blake runs a local taxi service. Lately his phone has been flooded with customer complaints. He gives you the following list of complaints. REQUIRED: a. Draw a Pareto chart of these problems....
-
The leading causes of death in low-income countries are listed in the table. Compare them with the leading causes of death in the United States given earlier in the chapter. Leading Causes of Death...
-
The following processes constitute the air-standard Diesel cycle: 12: isentropic compression,23: constant-volume energy addition (T and P increase),34: constant-pressure energy addition (v...
-
Convert metric units 8.55 g to mg
-
A friend owes you $500 in 1 year from now, and then $1000 2 years from now. Both loans are at 5%. If you want to replace both of these loans with ONE PAYMENT NOW, how much would your friend give you ?
-
If the amount of retained earnings at the beginning of the year was $ 3 0 , 0 0 0 , and $ 1 2 , 0 0 0 in dividends is paid during the year, calculate net income for the year.
-
On January 2, 20Y4, Whitworth Company acquired 33% of the outstanding stock of Aloof Company for $330,000. For the year ended December 31, 20Y4, Aloof Company earned income of $86,000 and paid...

Study smarter with the SolutionInn App


