A fluid obeys the Clausius equation of state where b(T) = b 0 + b 1 T.
Question:
A fluid obeys the Clausius equation of state
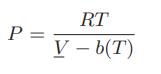
where b(T) = b0 + b1T. The fluid undergoes a Joule Thomson expansion from T1 = 120.0°C and P1 = 5.0 MPa to a final pressure P2 = 1.0 MPa. Given that CP = 20.97J/(mol K), b0 = 4.28 × 10−5 m3/mol, and b1 = 1.35 × 10−7 m3/(mol K), determine the final temperature of the fluid.
Transcribed Image Text:
P = RT V-b(T)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To determine the final temperature of the fluid we can use the following steps Calculate the enthalpy change for the JouleThomson expansion The enthal...View the full answer

Answered By

Vamsi Krishna Reddy Munnangi
Currently graduating in Computer Science and Engineering from UCEK JNTUK Kakinada, the best college in Andhra Pradesh,India.
TUTORING EXPERIENCE:
1)Worked with students aged 7-13 to improve Math, English, and Reading skills.
2)Reviewed materials assigned by the students' teachers and helped students complete homework.
3)Created lesson plans, review worksheets, and practice tests to prepare students for weekly tests.
4)Prepared students who successfully passed the LEAP test.
5)Communicated with parents about students' progress.
6)Successfully made three groups of grade D students acquire grade B levels in mock examinations.
7)Achieved 100% client satisfaction while scheduling and teaching independently.
8)Tutored many Computer Science students and developed programming skills in them from scratch.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The Clausius equation of state is a. Show that for this volumetric equation of state b. For a certain process the pressure of a gas must be reduced from an initial pressure P 1 to the final pressure...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
A bond has an expected return of 15.33%, sensitivity to the term factor of 1.95 and to the default factor of 0.65. The current term risk premium is 3.01%. The risk-free rate of return is 3%. What is...
-
When considering an application for a special use permit to develop and operate a ski resort at Sandy Butte, a mountain in Washington that is part of a national forest, the Forest Service prepared an...
-
Two types of instruments for measuring the amount of sulfur monoxide in the atmosphere are being compared in an air-pollution experiment. The following readings were recorded daily for a period of 2...
-
Five projects form the mutually exclusive, collectively exhaustive set under consideration. The cash flow profiles for the five projects are given in the table below. Information on each project was...
-
Mt. Sinai Hospital in New Orleans is a large, private, 600-bed facility, complete with laboratories, operating rooms, and x-ray equipment. In seeking to increase revenues, Mt. Sinai's administration...
-
1. An object of mass of 4 kg is resting at a height of 2 m and then falls to the ground. a. Model the energy of the object-Earth system with Energy Bar Charts when at the initial height and ground...
-
Show that the requirement for stability of a closed system at constant entropy and pressure leads to the condition that C P > 0 for all stable fluids.
-
The vant Hoff corollary to the third law of thermodynamics is that whenever two solid forms of a substance are known, the one with the greater specific heat will be the more stable one at higher...
-
How is the cost of capital decided upon.
-
Why is Singer's proposal to identify death by his specified cognitive function still run into problems? It wouldn't have settled the issue in Terry Schiavo's case. 'Death' is a biological concept,...
-
What does hikikomori mean? Group of answer choices suicide by gunshot social withdrawal to one's residence for six months irrational jealousy over learning about a partner's past a slow form of...
-
Who do you think will win the appeal? Why? What do you believe is the most persuasive argument of Baker? Of the State? Are there any issues with the briefs' writing/editing/proofreading? If so, tell...
-
"Change is a psychological process where people gradually accept the details of the new situation and the changes that come with it." This statement is a part of which model? a. Transition Model b....
-
Two HR managers are discussing how to manage job satisfaction in the workplace. One HR manager claims that organisations can manage job satisfaction using both job design and personnel selection ....
-
What is a VPN?
-
United Business Forms capital structure is as follows: Debt ............................................ 35% Preferred stock ........................... 15 Common equity .......................... 50...
-
A brick wall 3 m high, 7.5 m wide, and 200 mm thick has a thermal conductivity of 0.7 W/(m C). The temperature on the inner face is 25C, and the temperature on the outer face is 0C. How much heat is...
-
A 2500-lb automobile comes to a complete stop from 65 mph. If 60% of the braking capacity is provided by the front disk brake rotors, determine their temperature rise. Each of the two cast-iron...
-
A small hydroelectric power plant operates with 500 gal of water passing through the system each second. The water falls through a vertical distance of 150 ft from a reservoir to the turbines....
-
Let X = II{0, 1} be the infinite product space, equipped with the measure , where is a given fixed probability on the finite set {0,1}. Let & be the exchangeable -algebra of all measurable sets...
-
The Big Mac Index demonstrates the concept of Purchasing Power Parity. If the current price of a Big Mac hamburger in the United States is $3.80, and the current price of the same burger in Germany...
-
1. Identify any outliers in the following sets of data and explain your choices. a) X 25 34 43 55 92 105 16 Y 30 41 52 66 18 120 21 b) X 5 7 6 6 4 8 Y 304 99 198 205 106 9 2. a) Perform a linear...

Study smarter with the SolutionInn App


