Prove that since total mass is conserved during a chemical reaction, where m i is equal to
Question:
Prove that since total mass is conserved during a chemical reaction,

where mi is equal to the molecular weight of species i. Also show, by direct substitution, that the first of these equations is satisfied for the reaction
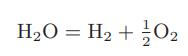
Transcribed Image Text:
and i=1 v₁m₁ = 0 for a single-reaction system Σvim; = i=1 Vijm; = 0 j = 1,2,..., M for a multiple-reaction system
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Lets examine the provided statements and the given reaction H2O H2 O2 to illustrate the conservation ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
Below is a list of the ingredients for Joanne Chang's yellow birthday cake as described in her cookbook, Flour. 2/3 cup buttermilk 1 cup butter, softened 1/3 cups sugar 2 large eggs (35g each) 2/....
-
The contamination of water-saturated soils with toxic organic solvents is an important environmental problem. The organic solvents can dissolve into to the water and diffuse through the...
-
Consider the chemical equilibrium of a solution of linear polymers made up of identical units. The basic reaction step is monomer + Nmer = (N + 1)mer. Let K N denote the equilibrium constant for this...
-
Consider the following limit order book for a stock.The last trade in the stock occurred at a price of $55. Buy Order Limit Sell Order Limit Precio share Precio share ps 54.75 500 ps 55.25 200 54.50...
-
Victor sells to Bonnie a refrigerator for $600 payable in monthly installments of $30 for twenty months. Bonnie signs a security agreement granting Victor a security interest in the refrigerator. The...
-
Find the mean, median, and mode for the sample whose observations, 15, 7, 8, 95, 19, 12, 8, 22, and 14, represent the number of sick days claimed on 9 federal income tax returns. Which value appears...
-
Quilts R Us (QRU) is considering investing in a new patterning attachment with the cash flow profile shown in the table below. QRU's MARR is 13.5 percent/year. a. What is the internal rate of return...
-
Hazlett, Inc., operates at capacity and makes plastic combs and hairbrushes. Although the combs and brushes are a matching set, they are sold individually and so the sales mix is not 1: 1. Hazletts...
-
Database design is quite complex and important. Discuss the role played by users during the design process Describe the purpose of normalizing data Briefly describe the basic SQL DDL statements and...
-
Show that the criterion for chemical equilibrium developed in the text, for a closed system at constant temperature and pressure, is also the equilibrium condition to be satisfied for closed systems...
-
In Sec. 8.7 we established that the condition for equilibrium between two phases is for closed systems either at constant temperature and pressure or at constant internal energy and volume. Show that...
-
Repeat Example 5.2 with a solvent for which E = 0.90. Display your results in a plot like Figure. Does countercurrent flow still have a marked advantage over crosscurrent flow? Is it desirable to...
-
In March 2011, Fukushima Japan faced a tripartite catastrophe: an earthquake, a tsunami and a nuclear meltdown. If you were responsible for public information outline how you would have managed both...
-
Then consider an older commercial, before social media was (as big) an influence. You can look at famous ads on Youtube. In a minimum of 3 pages, please do the following: Provide an introduction with...
-
Issue Specific Security Policies Locate an ISSP for a company you are interested in working for that you consider is either out of date, unfair, or ineffective in todays information age. Discuss why...
-
Read the following articles surrounding the organizational structure, leadership, and strategy. You will answer the following questions: Today, many organizations do not use formal organizational...
-
What are embedded audit modules? What is computer audit software or generalized audit software? How is a financial audit different from an information systems audit?
-
Typically, which is more difficult to detect and more costly to fixa design flaw in the processes or a programming error?
-
Question 2 For an n x n matrix A = form) via (aij)
-
Find an example of a structure or machine that has several forces acting on it. (a) Make a clear, labeled drawing of it. (b) Estimate the dimensions and the magnitudes and directions of the forces...
-
Many worldwide cities are in locations where extreme weather conditions can damage critical urban infrastructures. Select a representative at-risk city and design a structural system to shield the...
-
Find a real physical example of a mechanical structure or machine that has tensile stress present. (a) Make a clear, labeled drawing of the situation. (b) Estimate the dimensions of the structure or...
-
Training Plan for Conflict Resolution... Provide References Introduction: The training program will begin with an overview of the importance of conflict resolution and its impact on the workplace....
-
What are the considerations for implementing advanced concurrency control mechanisms, such as optimistic concurrency control, snapshot isolation, and Serializable Snapshot Isolation (SSI), to ensure...
-
Describe the components of the team-building cycle and why it is important to understand each phase. In addition, select one of the six stages and provide a personal/professional example of how you...

Study smarter with the SolutionInn App


