Show that the criterion for chemical equilibrium developed in the text, for a closed system at constant
Question:
Show that the criterion for chemical equilibrium developed in the text,
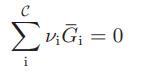
for a closed system at constant temperature and pressure, is also the equilibrium condition to be satisfied for closed systems subject to the following constraints:
a. Constant temperature and volume
b. Constant internal energy and volume
Transcribed Image Text:
C ΣvG = 0 i
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The image youve provided shows the mathematical condition for chemical equilibrium where i represents the stoichiometric coefficient for each substance and i represents the partial molar Gibbs free en...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
A student is using a model to show how charge is induced. A balloon that was rubbed on animal fur and charged negatively is brought near two aluminum cans resting on two separate cups. Draw (-) signs...
-
In Sec. 8.7 we established that the condition for equilibrium between two phases is for closed systems either at constant temperature and pressure or at constant internal energy and volume. Show that...
-
a. Show that the condition for equilibrium in a closed system at constant entropy and volume is that the internal energy U achieve a minimum value subject to the constraints. b. Show that the...
-
Establish procedures to guarantee substantiation of claims for allowances. Think about how you would set up a process to ensure employee claims and allowances could be claimed. Write a step by step...
-
On January 2, Burt asked Logan to loan him money against my diamond ring. Logan agreed to do so. To guard against intervening liens, Logan received permission to file a financing statement, and Burt...
-
Taxable income and pretax financial income would be identical for Huber Co. except for its treatments of gross profit on installment sales and estimated costs of warranties. The following income...
-
The accounting records for ADR, Inc., reflected the following amounts at the end of August 2010: Prepare ADRs multi-step income statement for the fiscal year ended August 31,2010. Cash...... $3,500...
-
Columbia Paper has the following stockholders equity account. The firms common stock has a current market price of $30 per share. Preferred stock .............$100,000 Common stock (10,000 shares at...
-
How to calculate rent if R15 000 is for the whole building but factory is 7 m, administration 2 m, office 1 m
-
Write a program that reads a connected graph from a file. The graph is stored in a file using the same format specified in Programming Exercise. Your program should prompt the user to enter the name...
-
Show that the partial molar volumes computed from Eqs. 8.6-4a and b and the partial molar enthalpies computed from Eqs. 8.6-9a and b must satisfy the Gibbs-Duhem equation. Amix V-1 a(Amix V) 1 T,P...
-
Prove that since total mass is conserved during a chemical reaction, where m i is equal to the molecular weight of species i. Also show, by direct substitution, that the first of these equations is...
-
Of the following, the one that is a BrnstedLowry acid is (a) [Cu(NH 3 ) 4 ] 2+ ; (b) [FeCl 4 ] ; (c) [Fe(H 2 O) 6 ] 3+ ; (d) [Zn(OH) 4 ] .
-
The current asset conversion cycle is the number of days it takes to purchase, produce, and sell an item, and then to collect on that item. Explain
-
(5) The failure time for a medical device, X, has a spliced distribution. (i) f 1 (x) follows the uniform distribution on (0,3). (ii) f 2 (x) follows the exponential distribution with mean 4. a 1 f 1...
-
Calculate MIRR for the project cash flows below using the "combination approach" as used in the MindTap homework set. The firm has a WACC of 10%. Time 0 1 2 3 Cash Flow - $ 500 $ 600 $ -400 $ 500
-
Prepare the Form 1040 by given the table below. Other income: 1099-INT Old Bank 2,300 1099-DIV Bake Co., Inc.-Ordinary dividends 3,100 Qualified dividends 3,100 Cassandra had the following itemized...
-
In the following frequency response diagram, find BW and fe (n MHz): OdB -3dB BW fe f (MHz)
-
What characteristics constitute a good fraud risk assessment?
-
Question 2 For an n x n matrix A = form) via (aij)
-
Ball bearings support a shaft at points A and B (Figure P4.30). The shaft is used to transmit power between two V-belts that apply forces of 1 kN and 1.4 kN to the shaft. Determine the magnitudes and...
-
A portable music player is sitting in a docking station (Figure P4.31). The docking station has a mass of 500 g and the player, 100 g. Determine the reaction forces at the two supports. Figure P4.31...
-
Two pots of food are being cooked on a solar cooker (Figure P4.32). The smaller pot weighs 4 lb, and the larger pot weighs 9 lb. Also, due to the thermal expansion of the parabolic reflector, a...
-
6. Let G be a CFG with the following rules: SaSble Let Li = {ab" In 20}. Prove that L CL(G).
-
Shamrock, Inc.reports the following liabilities (in thousands) on its December 31, 2022, balance sheet and notes to the financial statements. Accounts payable $4,701 Mortgage payable $7,006 Unearned...
-
Contrast the Universal and Contingency Models of strategy development. Explain the key differences between these two approaches, highlighting their respective strengths and weaknesses. Additionally,...

Study smarter with the SolutionInn App


