Starting with an equimolar mixture of methanol and NAD + at 25C, calculate and plot the extent
Question:
Starting with an equimolar mixture of methanol and NAD+ at 25°C, calculate and plot the extent of the following reaction as a function of pH:
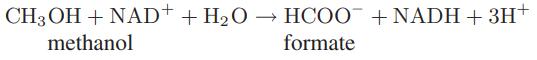
For this reaction at solution conditions, the apparent Gibbs energy change is ΔrxnG = −15.1 kJ. Also, assume that water is present in great excess, so that its concentration does not change in the course of the reaction.
Transcribed Image Text:
CH3OH + NAD+ + H₂O → HCOO + NADH + 3H+ methanol formate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
To calculate and plot the extent of the given reaction as a function of pH we need to use the Nernst ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
Starting with an equimolar mixture of acetaldehyde and NAD + at 25C, calculate and plot the extent of the following reaction as a function of pH. Also determine the pH at which half the acetaldehyde...
-
Starting with an equimolar mixture of lactate and NAD+ at 25C, calculate and plot the extent of the following reaction as a function of pH: where R is a side group. For this reaction at solution...
-
An equimolar mixture of oxygen and nitrogen enters a compressor operating at steady state at 10 bar, 220 K with a mass flow rate (m) of 1 kg/s. The mixture exits the compressor at 60 bar, 400 K with...
-
It is reported that 85% of Asian, 78% of white, 70% of Hispanic, and 38% of black children have two parents at home. Suppose there are 500 students in a representative school, of which 280 are white,...
-
Describe the two-stage allocation process for assigning support service costs to products in a traditional manufacturing environment.
-
An accounts clerk has compiled Trent's trial balance as at 31 March 2010 as follows: Required: Compile Trent's corrected trial balance as at 31 March2010. Dr Cr Bank (overdrawn) Capital Discounts...
-
In a study of reaction times, the time to respond to a visual stimulus (x) and the time to respond to an auditory stimulus (y) were recorded for each of 10 subjects. Times were measured in...
-
The comparative balance sheet of Morston Educational Supply at December 31, 2016, reported the following: Morstons transactions during 2016 included the following: Requirements 1. Prepare the...
-
Volte Corporation produces small electric appliances. The following information is available for the most recent period of operations: Standard variable overhead rate Actual output Actual direct...
-
Determine the charge on tyrosine as a function of pH. Tyrosine (C 9 H 11 NO 3 ), another amino acid in proteins, has two dominant ionizable groups with pK HA values of 2.24 and 9.04 at 25C (and a...
-
Adenosine monophosphate (AMPH) is a nucleotide that is present as a monomer in DNA and RNA. It consists of a phosphate group, a ribose molecule and an adenine molecule. Consequently, it can be found...
-
True or False: 1. There is virtually no positive correlation between the earnings of grandparents and grandchildren. 2. Other things being equal, workers who prefer more amenities at work or more...
-
Ramos Company has the following unit costs: Variable manufacturing overhead Direct materials Direct labor Fixed manufacturing overhead Fixed marketing and administrative $19 18 23 16 14 What cost per...
-
Gardner Sail Makers manufactures sails for sailboats. The company has the capacity to produce 36,000 sails per year and is currently producing and selling 30,000 sails per year. The following...
-
Consider a rm that only has a patent as its assets If not developed any further, the value of the patent will only be $19 million at the end of the year. The rm has long-term debt of $28 million,...
-
If you were one of the original investors in this issue and you had invested $10,000, what would your total return be if you sold the securities at today's market price? Compute the current duration...
-
A coaxial cable consists of a very long hollow cylindrical wire, containing a long solid cylindrical wire. The cross- section of the cable is shown to the right. The inner wire has a radius of R, and...
-
Advanced Vehicle Enterprises (AVE) follows a policy of paying out 50% of its net income as cash dividends to its shareholders each year. The company plans to do so again this year, during which AVE...
-
Les has collected stamps in his spare time for years. He purchased many of his stamps at a price much lower than the current market value. Les recently lost his job as a carpenter. Since his wife...
-
The following figure is an exploded view of a box. Identify and label all the components. TT T T T T
-
Draw a conceptual sketch of an incandescent light bulb. Identify all the components using arrows and numbers as in Figure 1.1. Figure 1.1 11 Radiator: Battery: 10 Spare Tire: Exhaust Manifold: 9. Gas...
-
There are 39 inches in a meter. What is the volume in the Engineering English system of a spherical apple that is 10. cm (note the decimal point here) in diameter? Assume that on the surface of the...
-
local health care facility has fixed costs per month of $187 400. They also have patient costs of $4.15 per day per patient for linen and cleaning, medication costs are $23.32 per patient per day and...
-
Products is a cranberry cooperative that operates two divisions, a harvesting division and a processing division. Currently, all of harvesting's output is converted into cranberry juice by the...
-
Five people get into a car with an air exchange rate of 4 h-1 and an interior volume of 4 m3 . Assume each person emits 35 g CO2/h. The five people drive for 2 hours total. a. Calculate the CO2...

Study smarter with the SolutionInn App


