The following set of reactions is thought to occur between nitrogen and oxygen at high temperatures a.
Question:
The following set of reactions is thought to occur between nitrogen and oxygen at high temperatures
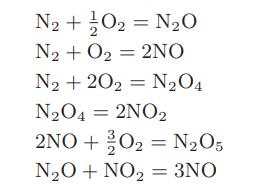
a. Find an independent set of reactions for the nitrogen-oxygen system.
b. How many degrees of freedom are there for this system?
c. If the starting oxygen-to-nitrogen ratio is fixed (as in air), how many degrees of freedom are there?
Transcribed Image Text:
N2+}O2 = N2O N₂ + O₂ = 2NO N₂ +202 N₂O4 N₂O4 = 2NO2 2NO+20₂ = N₂O5 N₂O + NO₂ = 3NO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The following set of reactions is thought to occur between nitrogen and oxygen at high temperatures ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The following set of questions relates to using Poisson regression methods to analyze data from an in vitro study of human chromosome damage. In this study, using Poisson regression is appropriate...
-
Ann Carter, Chief Financial Officer of Consolidated Electric Company (Con El), must make a recommendation to Con Els board of directors regarding the firms dividend policy. Con El owns two...
-
Ivan Petrov was a member of the newly formed entrepreneur class in Bulgaria; partly out of desire, mostly out of necessity. The changes occurring as the country moved from a centrally planned to a...
-
On December 31, Alvare Company estimates its bad debts to be .5% of its annual credit sales of $875,000. They use the allowance method. Prepare the Journal entries for December 31 recording the Bad...
-
Jones bought a used car from the AHerts Car Rental System, which regularly sold its used equipment at the end of its fiscal year. First National Bank of Roxboro had previously obtained a perfected...
-
Loxley Company has the following portfolio of investment securities at September 30, 2008, its last reporting date. On October 10, 2008, the Fogelberg shares were sold at a price of $54 per share. In...
-
The following transactions occurred during April 2010, for Angelos Garden Center, Inc.: Requirement 1. Journalize the April transactions for Angelos Garden Center, Inc. Omit explanations. Apr 3...
-
Apply the value chain model to a video game developer, such as Electronic Arts (www.EA.com). What is its competitive strategy? Describe the tasks Electronic Arts must accomplish for each of the...
-
Regina Company has the following balances in its overhead accounts at the end of the year: Manufacturing Overhead, Control Manufacturing Overhead Allocated $ 486,000 480,000 The balance of several...
-
A firm is considering replacing a machine that has been used to make a certain kind of packaging material. The new, improved machine will cost $31,000 installed and will have an estimated economic...
-
The molar integral heat of solution s H is defined as the change in enthalpy that results when 1 mole of solute (component 1) is isothermally mixed with N 2 moles of solvent (component 2) and is...
-
a. What is the maximum number of phases that can coexist for a mixture of two nonreacting components? b. How would the answer in part (a) change if the two components could react to form a third...
-
Factor the trinomial. 8x 2 - 2x - 3
-
1. Mr. JM Tanvir has deposited Tk 15,000 in a bank at 10% annual interest on 31st December 2017. 2. What amount will he receive on 1st January 2022 if the bank charge interest quarterly basis?
-
A customer has cargo to be shipped by air from Toronto (YYZ) to Japan (NRT). There are15 cartonsglass goblets, packed 12 per carton, in this shipment. The dimensions of each carton areL60cm xW30cm x...
-
A tour operator believes that the profit P , in dollars, from selling x tickets is given byP ( x ) = 4 5 x - 0 . 2 5 x ^ 2 . Using this model, what is the maximum profit the tour operator can expect?
-
1. Develop a balanced scorecard for your own life. What are your personal, professional or academic goals? Be sure to document each step shown in the Balanced Scorecard video, specifically addressing...
-
Otto purchased a new car from Dealer for $20,000. He paid for the car by borrowing $15,000 from Dealer. After a few months, Otto realized there were some serious flaws with the car--he kept having to...
-
Why is it important that management and auditors collaborate on a fraud risk assessment?
-
What are the two methods used to translate financial statements and how does the functional currency play a role in determining which method is used?
-
The three forces (magnitudes 100 lb, 200 lb, and P) combine to produce a resultant R (Figure P4.9). The three forces act in known directions, but the numerical value P is unknown. (a) What should the...
-
Find a real physical example of a mechanical structure or machine that has a moment acting on it. (a) Make a clear, labeled drawing of the situation. (b) Estimate the dimensions, and the magnitudes...
-
Resulting from a light wind, the air pressure imbalance of 100 Pa acts across the 1.2-m by 2-m surface of the highway sign (Figure P4.11, see on page 172). (a) Calculate the magnitude of the force...
-
Calculate the firm's WACC (using 2018 numbers). (You will need to collect information on the long-term debt and common stock equity from the Balance Sheet. The firm has no preferred stock). Use the...
-
You need to borrow $10,000,000 on your line of credit to cover increased working capital expense due to sales growth. Bank A is willing to lend to you at 5.25% with 10% compensating balances. Bank B...
-
Compute the consolidated assets in 2023 Finale Corporation acquired 80% of the outstanding shares of Closing Company on January 2, 2022 for P3,250,000, excluding control premium of P125,000. On this...

Study smarter with the SolutionInn App


