The process for making cyclohexane by saturation of benzene consists of a feed-effluent heat exchanger, a saturation
Question:
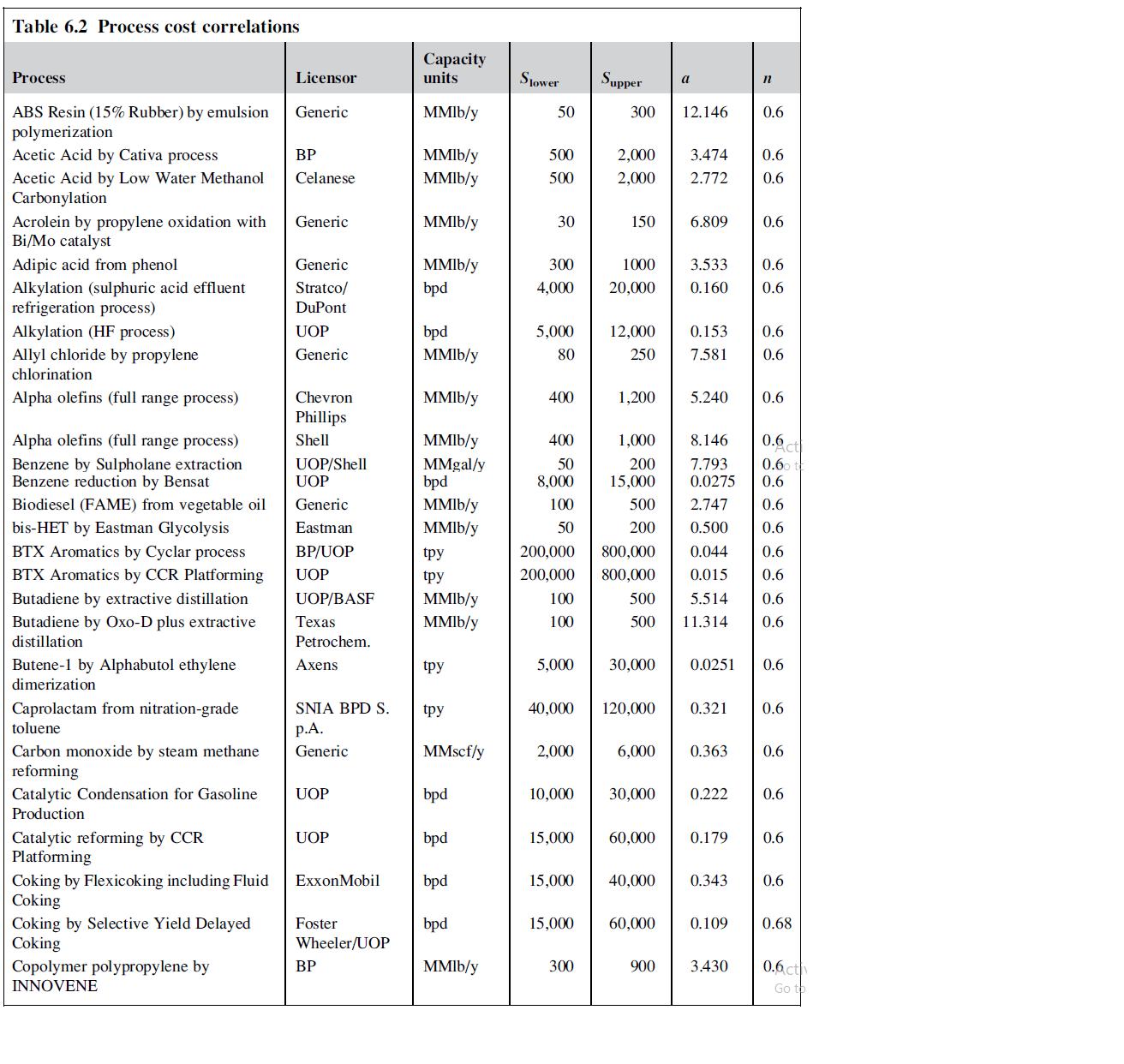
The process for making cyclohexane by saturation of benzene consists of a feed-effluent heat exchanger, a saturation reactor and a product stabilizer column. Estimate the cost of a plant that produces 200,000 metric tons per year (200 kte/y) of cyclohexane using the correlation in Table 6.2 and Bridgewater’s method.
Transcribed Image Text:
Table 6.2 Process cost correlations Process ABS Resin (15% Rubber) by emulsion polymerization Acetic Acid by Cativa process Acetic Acid by Low Water Methanol Carbonylation Acrolein by propylene oxidation with Bi/Mo catalyst Adipic acid from phenol Alkylation (sulphuric acid effluent refrigeration process) Alkylation (HF process) Allyl chloride by propylene chlorination Alpha olefins (full range process) Alpha olefins (full range process) Benzene by Sulpholane extraction Benzene reduction by Bensat Biodiesel (FAME) from vegetable oil bis-HET by Eastman Glycolysis BTX Aromatics by Cyclar process BTX Aromatics by CCR Platforming Butadiene by extractive distillation Butadiene by Oxo-D plus extractive distillation Butene-1 by Alphabutol ethylene dimerization Caprolactam from nitration-grade toluene Carbon monoxide by steam methane reforming Catalytic Condensation for Gasoline Production Catalytic reforming by CCR Platforming Coking by Flexicoking including Fluid Coking Coking by Selective Yield Delayed Coking Copolymer polypropylene by INNOVENE Licensor Generic ВР Celanese Generic Generic Stratco/ DuPont UOP Generic Chevron Phillips Shell UOP/Shell UOP Generic Eastman BP/UOP UOP UOP/BASF Texas Petrochem. Axens SNIA BPD S. p.A. Generic UOP UOP ExxonMobil Foster Wheeler/UOP BP Capacity units MMlb/y MMlb/y MMlb/y MMIb/y MMlb/y bpd bpd MMlb/y MMIb/y MMIb/y MMgal/y bpd MMlb/y MMlb/y tpy tpy MMIb/y MMIb/y tpy tpy MMscf/y bpd bpd bpd bpd MMIb/y Slower 50 500 500 30 300 4,000 5,000 80 400 400 50 8,000 100 50 5,000 2,000 15,000 Supper 15,000 300 15,000 2,000 2,000 300 150 1000 20,000 200,000 800,000 200,000 100 100 12,000 250 1,200 1,000 200 15,000 40,000 120,000 500 200 30,000 6,000 60,000 40,000 a 60,000 12.146 8.146 0.6 7.793 0.6 0.0275 0.6 2.747 0.6 0.500 0.6 0.044 0.6 0.6 800,000 500 500 11.314 0.6 0.015 5.514 0.6 900 6.809 10,000 30,000 0.222 3.474 0.6 2.772 0.6 5.240 3.533 0.6 0.160 0.6 0.153 0.6 7.581 0.6 n 0.363 0.6 0.0251 0.6 0.179 0.6 0.321 0.6 0.343 0.109 3.430 0.6 0.6 0.6 0.6 0.6 0.68 0.6 ct Go t
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
From Table 62 the cost correlation for the Axens process for benzene saturation gives That we have obtained two very different answers Bridgewaters co...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Engineering Design
ISBN: 9780081025994
6th Edition
Authors: Ray Sinnott, R.K. Sinnott, Sinnott Gavin Towler
Question Posted:
Students also viewed these Engineering questions
-
Formaldehyde is produced from methanol in a catalytic oxidation reactor. The following reactions take place: CH3OH HCHO + H2 2H2 + O2 2H2O A stream of methanol is joined by a recycle stream, also...
-
A plant is required to produce 10,000 metric tons per year of anhydrous hydrogen chloride from chlorine and hydrogen. The hydrogen source is impure: 90 mol% hydrogen, balance nitrogen. The chlorine...
-
A catalytic process was designed to make 150 metric tons per year of product with a net profit of $0.25/lb of product. The catalyst for the process costs $10/lb and it takes 2 months to shut down the...
-
A metallic block (specific heat 500 J/kg K) of 100 kg mass having a temperature of 360 K is immersed in an insulated tank containing 40 kg of water at 300 K. Find the change in entropy of the...
-
What lessons, if any, can be learned from examining the history of foreign currency translation in the United States?
-
In Year 1, Lee Inc. billed its customers $62,000 for services performed. The company collected $51,000 of the amount billed. Lee incurred $39,000 of other operating expenses on account. Lee paid...
-
In addition to the customary horizontal velocity components of the air in the atmosphere (the "wind"), there often are vertical air currents (thermals) caused by buoyant effects due to uneven heating...
-
The following refers to units processed in Sunflower Printings binding department in March. Prepare a physical flow reconciliation. Units of Percent of Product Conversion Added Beginning work in...
-
Here is some hypothetical testimony from prosecution of an assault of a victim in a grocery store parking lot: The prosecutor calls Bart (an eyewitness and the "primary" or "fact" witness in this...
-
The process used in the manufacture of aniline from nitrobenzene is described in Appendix F, design problem F.8. The process involves six significant stages: Vaporization of the nitrobenzene...
-
A polymer is produced by the emulsion polymerization of acrylonitrile and methyl methacrylate in a stirred vessel. The monomers and an aqueous solution of catalyst are fed to the polymerization...
-
California and New York lead the list of average teachers salaries. The California yearly average is $64,421 while teachers in New York make an average annual salary of $62,332. Random samples of 45...
-
How would you define corruption as it applies to criminal justice and security organizations? What are the causes and implications of corruption? What preventative strategies are used against...
-
What is crypto, what is cryptography? What are the technologies behind it? ( example blockchain etc etc) what are the different types of Cryptocurrency? What is the market cap of cryptocurrency,...
-
What processes were employed and why / how did they contribute to operating success for the case study?
-
If a stock has a variance of 0.00192 and an expected rate of return of 14%, what is the standard deviation for this stock? Do not round intermediate calculations. Round the final answer to 2 decimal...
-
Think of 30 objects, phrases and experiences that relate to the product / strategic message. Then, try and twist one. Create an ad that has a twisted visual with a straight headline - or a straight...
-
Why does audit risk create business risk for an auditor, and how does the legal environment of business affect the auditors business risk?
-
A spacecraft has left the earth and is moving toward Mars. An observer on the earth finds that, relative to measurements made when the spacecraft was at rest, its a. length is shorter b. KE is less...
-
What is the difference between a false positive and a false negative?
-
The U.S. Department of Agriculture provided homogenized baby food samples to three labs for analysis.3 Results agreed well for protein, fat, zinc, riboflavin, and palmitic acid. Results for iron were...
-
The U.S. Department of Agriculture provided homogenized baby food samples to three labs for analysis.3 Results agreed well for protein, fat, zinc, riboflavin, and palmitic acid. Results for iron were...
-
An agency MBS pool is 46 months old, with an original size of $80m, and has a survival factor of 70%. The interest rate of the pool is 5%, and the pool was originally originated with 30-year term...
-
. Assume that consumers in a nation reduce their marginal propensity to save from 0.25 to 0.2 though their personal incomes initially do not change. How will this change in MPS affect the economy?...
-
At December 31, 2025, Sarasota Company has outstanding noncancelable purchase commitments for 39,500 gallons, at $3.42 per gallon, of raw material to be used in its manufacturing process. The company...

Study smarter with the SolutionInn App


