(a) Use the data in Appendix 2B to decide which of ozone and fluorine is the stronger...
Question:
(a) Use the data in Appendix 2B to decide which of ozone and fluorine is the stronger oxidizing agent in water.
(b) Does your answer depend on whether the reaction is carried out in acidic or basic solution?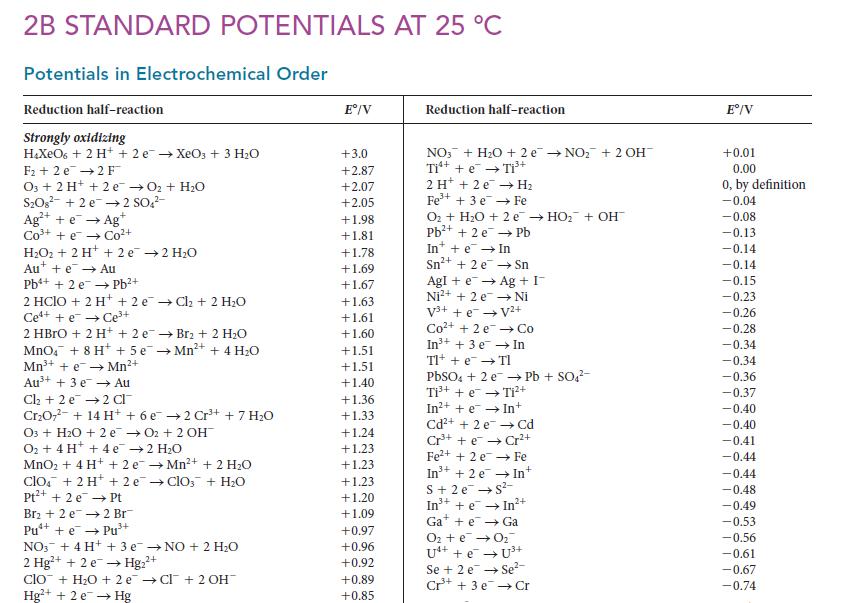
Transcribed Image Text:
2B STANDARD POTENTIALS AT 25 °C Potentials in Electrochemical Order Reduction half-reaction Strongly oxidizing H₂XeO6 + 2 H+ + 2 e → XeO3 + 3 H₂O F₂2 e 2 F™ 03 + 2 H+ + 2 e → O₂ + H₂O S₂O8² +2e →2 SO4²- Ag+ + e → Agt Cơ từ cot H₂O₂+ 2 H+ + 2e →2 H₂O Aue Au Pb+ + 2 e Pb²+ → 2 HClO + 2 H+ +2e →Ch₂ + 2 H₂O Ce++eCe³+ 2 HBrO + 2 H+ + 2 e MnO4 + 8 H+ + 5e Mn³+ + e→→Mn²+ Au³+ + 3 e Au Cl₂ +2 e 2 Cl Cr₂O7² + 14 H+ + 6 e 03 + H₂O + 2 e O₂ + 4 H+ + 4e MnO₂ + 4H+ + 2e clo + 2H+ + 2e Pt²+ + 2 e →→ Pt Br₂ + 2 e2 Br¯ Pu²+ + e→Pu³+ 3+ → Br2 + 2 H₂O →Mn²+ + 4 H₂0 2 Cr³+ + 7 H₂O 02 + 2 OH™ 2 H₂O →Mn²+ + 2 H₂O →ClO3 + H₂O NO3 + 4 H+ + 3 e→NO + 2 H₂O 2+ 2 Hg²+ + 2e → Hg₂²+ clo + H₂O +2e →Cl + 2 OH™ Hg²+ + 2 e → Hg Eº/V +3.0 +2.87 +2.07 +2.05 +1.98 +1.81 +1.78 +1.69 +1.67 +1.63 +1.61 +1.60 +1.51 +1.51 +1.40 +1.36 +1.33 +1.24 +1.23 +1.23 +1.23 +1.20 +1.09 +0.97 +0.96 +0.92 +0.89 +0.85 Reduction half-reaction NO3 + H₂O + 2e →NO₂+ 2 OH™ Ti¹+ + e → Ti³+ 2 H + 2e → H₂ Fe³+ + 3 e Fe O₂ + H₂O + 2 e → HO₂ + OH™ Pb²+ + 2e →→ Pb In+ + e → In Sn²+ + 2 e Sn Ag + I Ni Agi + e → Ni²+ + 2 e V³+ + e → V²+ Co²+ +2 e In³+ + 3 e Tl+ + e → TI Co In PbSO4 + 2 e Ti³+ + e → Ti²+ In²+ + e → In+ Cd²+ + 2e → Cd Cr³+ +eCr²+ Fe²+ + 2e → Fe In +2 e S+ 2e → S²- Pb + SO4²- + In Gae Ga O₂ + e → 0₂ U4+ + e → Se+ 2 e Cr³+ +eIn2+ Se²- + 3e → Cr Eº/V +0.01 0.00 0, by definition -0.04 -0.08 -0.13 -0.14 -0.14 -0.15 -0.23 -0.26 -0.28 -0.34 -0.34 -0.36 -0.37 -0.40 -0.40 -0.41 -0.44 -0.44 -0.48 -0.49 -0.53 -0.56 -0.61 -0.67 -0.74
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The data in Appendix 2B shows the standard potentials of various halfreactionsThe standard potential ...View the full answer

Answered By

Rustia Melrod
I am a retired teacher with 6 years of experience teaching various science subjects to high school students and undergraduate students. This background enables me to be able to help tutor students who are struggling with the science of business component of their education. Teaching difficult subjects has definitely taught me patience. There is no greater joy for me than to patiently guide a student to the correct answer. When a student has that "aha!" moment, all my efforts are worth it.
The Common Core standards are a useful yardstick for measuring how well students are doing. My students consistently met or exceeded the Common Core standards for science. I believe in working with each student's individual learning styles to help them understand the material. If students were struggling with a concept, I would figure out a different way to teach or apply that concept. I was voted Teacher of the Year six times in my career. I also won an award for Innovative Teaching Style at the 2011 National Teaching Conference.
4.90+
4+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Is it possible for a matrix to get out of control because of too many small projects, each competing for the same shared resources? If so, how many projects are too many? How can management control...
-
The Canada and Quebec pension system levies a tax on workers and pays benefits to the elderly. Suppose that government increases both the tax and the benefits. For simplicity, assume that the...
-
Use the data in Appendix 3 to calculate the equilibrium constant for the reaction Agl(s) Ag+(aq) + I2(aq) at 25C. Compare your result with the Ksp value in Table 16.2.
-
Why must Microsoft report this charge of over $1billion entirelyin one quarter, the last quarter of the company's physicalyear? From an accounting point of view, what willMicrosoft do in the futur?
-
Ms. Smith is retired and depends on her investments for retirement income. Mr. Jones is a young executive who wants to save for the future. They are both stockholders in Airbus, which is investing...
-
Sommer Graphics Company was organized on January 1, 2015, by Krystal Sommer. At the end of the first 6 months of operations, the trial balance contained the accounts shown below. Analysis reveals the...
-
Consider a payoff \(C\) that will occur in 2 years, taking one of the three possible values \(C_{0}, C_{1}, C_{2}\). The short rate lattice for these 2 years is shown in Figure 16.16, with \(d_{i...
-
Sentry, Inc. was started on January 1, Year 1. Year 1 Transactions 1. Acquired $20,000 cash by issuing common stock. 2. Earned $62,000 of revenue on account. 3. On October 1, Year 1, borrowed $12,000...
-
Bank Alpha has given out a pool of mortgages with principal amount of $13 million. These mortgages have an average maturity of 22 years and a monthly mortgage payment of 7 per cent per annum. (Assume...
-
Isoelectronic species have the same number of electrons. (a) Divide the following species into two isoelectronic groups: CN , N , NO 2 , C 2 2 , O 3 . (b) Which species in each group is likely to...
-
Classify each of the following hydrides as saline, molecular, or metallic: (a) LiH; (b) NH 3 ; (c) HBr; (d) UH 3 .
-
Determine the taxable income for the Columbia Corporation for the following independent cases: Income from STCG (NSTCL) NLTCG (NLTCL) Operations Case A B $110,000 100,000 $30,000 $44,000 65,000...
-
1. The following are two versions of Battle of Sexes: (a) A couple may go to the opera or to football. Each person will decide individually. If they both go to football, her payoff is 1, and his is...
-
Question 3: Using Python as a calculator We have seen how Python can be used to evaluate mathematical expressions. This problem provides practice incc constants. Write python code to do the...
-
Determine the embodied energy for the wall material (Ignore the energy requirements for mortar and construction process). Clay brick is used for construction of a 10 m wall as shown in figure....
-
(a) Estimate the time required (in units of gate delay) to perform 64-bit addition using cascaded 4-bit carry-lookahead units. (b) A BCD (Binary-Coded Decimal) number is a 4-bit representation with...
-
Write a program that reads two dates entered in the format " day.month.year " and calculates the number of days between them . Enter the first date: 27.02.2006 Enter the second date: 3.03.2006...
-
How are you, as an owner/manager, involved in behavior modeling?
-
The baseball player A hits the ball from a height of 3.36 ft with an initial velocity of 34.8 ft/s. 0.14 seconds after the ball is hit, player B who is standing 15 ft away from home plate begins to...
-
Why can the signal loss resulting from spin dephasing caused by magnetic field in-homogeneities and chemical shift be recovered in the spin-echo experiment?
-
Why do neighboring groups lead to a net induced magnetic field at a given spin in a molecule in the solid state, but not for the same molecule in solution?
-
Why is it useful to define the chemical shift relative to a reference compound as follows? 8 = 10- Vref) Vref
-
A copy machine cost $39,000 when new and has accumulated depreciation of $25,000. Suppose Print Center sold the machine for $14,000. What is the result of this disposal transaction?
-
Write named CHECK constraints for the customer balance is greater than or equal to zero and customer state is one of CO or CA in the CREATE table statement to add the named CHECK constraints ?
-
Explain the concept of 'organizational behaviour modification' with Example

Study smarter with the SolutionInn App


