At a given temperature, K = 1.3 10 -2 for the reaction N(g) + 3H(g)2NH(g) Calculate
Question:
At a given temperature, K = 1.3 × 10-2 for the reaction
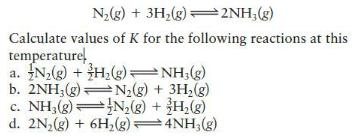
Transcribed Image Text:
N₂(g) + 3H₂(g)2NH₂(g) Calculate values of K for the following reactions at this temperature a. N₂(g) + H₂(g) →NH3(g) b. 2NH₂(g) c. NH3(g) d. 2N₂(g) + 6H₂(g) → 4NH3(g) N₂(g) + 3H₂(g) N₂(g) + 3H₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
Values of equilibrium constant at given temperature for the following reactions are 011 769 88 and 1...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Given K = 3.50 at 458C for the reaction A(g) + B(g) C(g) and K = 7.10 at 458C for the reaction 2A(g) + D(g) C(g) what is the value of K at the same temperature for the reaction C(g) + D(g) 2B(g)...
-
At 10 000 K the ionization reaction for Ar is: Ar Ar+ + e with equilibrium constant of K = 4.2 104. What should the pressure be for a mole concentration of argon ions (Ar+) of 10%?
-
A temperature of about 0.01 K can be achieved by magnetic cooling. In this process a strong magnetic field is imposed on a paramagnetic salt, maintained at 1 K by transfer of energy to liquid helium...
-
Assume an employee signs a non - disparagement clause. What would be considered breaking this clause? a . ) Telling a company's trade secret to a competing firm for a fee b . ) Using a work computer...
-
Verify the rows of the transition matrix in Example 3.10.6 that correspond to current states {AA, Aa} and {Aa, aa}.
-
What is the difference between a static budget and a flexible budget? When is each used?
-
Air at standard conditions flows through an 8-in.-diameter, 14.6-ft-long, straight duct with the velocity versus head loss data indicated in the following table. Determine the average friction factor...
-
The following preliminary unadjusted trial balance of Seats-For-You Co, a sports ticket agency, does not balance: When the ledger and other records are reviewed, you discover the following: (1) the...
-
What are the critical elements of a robust Management of Change (MoC) program in the context of PSM? Discuss how effective MoC practices contribute to minimizing risks associated with process...
-
On December 1, 2020, Cambridge Printers had the account balances shown below. Debit Credit Cash 14,650 Accumulated DepreciationEquipment 1,500 Accounts Receivable 3,900 Accounts Payable 3,000...
-
An equilibrium mixture contains 0.60 g solid carbon and the gases carbon dioxide and carbon monoxide at partial pressures of 2.60 atm and 2.89 atm, respectively. Calculate the value of K p for the...
-
Consider the same reaction as in Exercise 12. In a particular experiment, 1.0 mole of H 2 O(g) and 1.0 mole of CO(g) are put into a flask and heated to 3508C. In another experiment, 1.0 mole of H 2...
-
Euro Classics Ltd has been operating a successful business for many years specialising in servicing and reconditioning repairs for classic European automobiles. Servicing costs $750, while...
-
It is new Year even and the roads are icy. the coefficient of friction between your tires and the road is only 0.40. Compute the tightest radius curve you can negotiate while traveling on a level...
-
Marco Manufacturing has a stamping machine that originally cost $20,000 and still has one year of depreciation remaining to be charged on the machine. This machine creates an annual return of $10,000...
-
The tape in a videotape cassette has a total length 2 5 0 m and can play for 2 . 5 h . As the tape starts to play, the full reel has an outer radius of 3 5 mm and an inner radius of 1 0 mm . At some...
-
Gross profit, operating profit, and net profit; the three most important profitability ratios. Business owners and managers may find varied usefulness of the other ratios calculated from the income...
-
5) It costs a company c(x) = -0.004x2 + 500x + 50000 to make its product. Find the marginal cost and use it to find how fast the cost is increasing when x = 2000 units. (4 points)
-
Convert the [100] and [111] directions into the four-index Miller-Bravais scheme for hexagonal unit cells.
-
Research an article from an online source, such as The Economist, Wall Street Journal, Journal of Economic Perspectives, American Journal of Agricultural Economics, or another academic journal. The...
-
The gravitational force exerted by an object is given by F = mg where F is the force in newtons, m is the mass in kilograms, and g is the acceleration due to gravity, 9.81 m/ s 2 . Calculate the...
-
Consider two different containers, each filled with 2 moles of Ne(g). One of the containers is rigid and has constant volume. The other container is flexible (like a balloon) and is capable of...
-
An unknown diatomic gas has a density of 3.164 g/ L at STP. What is the identity of the gas?
-
Find the vertical asymptotes, if any, and the values of x corresponding to holes, if any, of the graph of the rational function.. f(x)= x-6 X-9x+18
-
on Jan 1 , mary took out a bank loan for $ 6 0 K with an interest rate of 5 % . Interest is payable on first of each following month. What is the posting for Sept 2 0 2 3 ?
-
Q3. Consider the expression below. (3) + (27) -2 Explain the mathematical properties you would use to simplify this expression into the form 3" and describe the process to find the value of a. A...

Study smarter with the SolutionInn App


