Calculate (Delta S_{text {surr }}) for the following reactions at (25^{circ} mathrm{C}) and (1 mathrm{~atm}). a. C3H8(g)
Question:
Calculate \(\Delta S_{\text {surr }}\) for the following reactions at \(25^{\circ} \mathrm{C}\) and \(1 \mathrm{~atm}\).
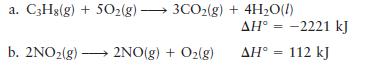
Transcribed Image Text:
a. C3H8(g) + 5O(g) 3CO2(g) + 4HO(l) b. 2NO2(g) 2NO(g) + O(g) AH = -2221 kJ AH = 112 kJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
The change in entropy of the surroundings Delta Stextsurr for a reaction can be determined by lookin...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate DG and Kc for the following reactions at 25C: (a) (b) (c) (d) 2 + Br2() 21 (aq)2Br (aq) I2(s) 02(8) 4H (a) 4Fe (aq) 2H20(1) + 4Fe (aq) 2.A l (s) + 312(s) 2A13+(aq) + 6(aq)
-
Calculate ÎSsurr for the following reactions at 25oC and 1 atm. a. C3Hsg) 502(g)3CO2ig) 4H20(D -2221 kJ ' '-112 kJ b, 2N02(g)-2N0(g) + O2(g)
-
The equilibrium constant of the reaction CO + ½ O2 CO2 at 1000 K and 1 atm is KP1 Express the equilibrium constant of the following reactions at 1000 K in terms of KP1: (a CO10CO at 3 atm at 1...
-
In your hometown what system is used to price the publicly supplied water? Why was that pricing system chosen? Would you recommend an alternative?
-
Consider the Henon map described by Let a = 1.4 and b = 0.3, and use a computer to plot the first 10,000 points (xw yn) starting from the initial values x0 = 0, y0 =0. Choose the plot region as 1.5 -...
-
For the orange-drink tank shown in Fig. 4.32, compute the magnitude and location of the total force on each vertical end wall. The tank is 3.0 m long. 30 Orange drink (sg - 1.10) 3.0 m 4.6 m 2.4-m...
-
Mr. Tam is the front man for the Asian American dance-rock band The Slants. Mr. Tam named his band The Slants to reclaim and take ownership of Asian stereotypes. The band draws inspiration for its...
-
Context-sensitive nature of cost behavior classifications Susan Hicks operates a sales booth in computer software trade shows, selling an accounting software package, Dollar System. She purchases the...
-
BHT Company evaluates a project for a potential inclusion in its capital budget. In the first year of the project, it expects incremental net income of $33 million, depreciation expenses of $8...
-
Given the following data: calculate \(\Delta G^{\circ}\) for the reaction \[6 \mathrm{C}(s)+3 \mathrm{H}_{2}(g) \longrightarrow \mathrm{C}_{6} \mathrm{H}_{6}(l)\] 2C6H6(l) +150(g) 12CO(g) + 6HO(l)...
-
Choose the compound with the greatest positional probability in each case. a. \(1 \mathrm{~mol}_{\text {of }} \mathrm{H}_{2}\) at STP or \(1 \mathrm{~mol}_{\text {of }} \mathrm{H}_{2}\) at...
-
Under which of the categories mentioned in Question 13.1 do XML documents fall? What about self-describing data?
-
Explain these features of legislation related to taxable transactions: 1. Taxable entities 2. Tax rates and schedules 3. Penalties
-
Draw an approximate diagram of the model, with capital per efficiency unit, ke, on the x-axis and output per efficiency unit, ye, on the y-axis (hint: your diagram should have three curves). Identify...
-
1. Identify the main characters in this case, and explain what happened. 2. To what extent did Lawson, Vandivier, and Gretzinger consider the relevant moral issues before deciding to participate in...
-
A company needs to sell a pillow to a specific customer at a lower price than other customers. You need to configure the discounted price to apply by default for all future orders. Where should you...
-
5. If your only income is salary paid by your employer, which of these statements about filing your taxes is true? * 1 point You need to file your tax return by the end of the calendar year...
-
Assume that the following events occurred at a division of Generic Electric for March of the current year. 1. Purchased $45 million in direct materials. 2. Incurred direct labor costs of $24 million....
-
Write electron configurations for the following ions, and determine which have noble-gas configurations: (a) Cd2+ (b) p3- (c) Zr4+ (d) Ru3+ (e) As3- (f) Ag+
-
The rate law of the reaction 2 NO(g) + 2 H 2 (g) N 2 (g) + 2 H 2 O(g) is Rate = kr[NO] 2 [H 2 ], and the mechanism that has been proposed is (a) Which step in the mechanism is likely to be rate...
-
(a) Use a graphing calculator or graphing software to calculate the activation energy for the acid hydrolysis of sucrose to give glucose and fructose (see Exercise 7.9) from an Arrhenius plot of the...
-
The decomposition of A has the rate law Rate = k r [A] a . Show that for this reaction the ratio t 1/2 /t 3/4 , where t 1/2 is the half-life and t 3/4 is the time for the concentration of A to...
-
the formula to compute the budgeted direct labor cost is Multiple choice question. units to produce times direct labor required per unit divided by direct labor cost per hour units to produce times...
-
Do you believe your organisation has a planned approach to career management? Motivate your answer What ought your employer do to manage your career? What ought you do yourself to contribute to your...
-
What service provider characteristic ) do consumers rely on to make purchase decisions for services whose quality is difficult to judge, even after purchasing?

Study smarter with the SolutionInn App


