Calculate the concentrations of Ag + , Ag(S 2 O 3 ) , and Ag(S 2
Question:
Calculate the concentrations of Ag+, Ag(S2O3)–, and Ag(S2O3)23– in a solution prepared by mixing 150.0 mL of 1.00 × 10–3 M AgNO3 with 200.0 mL of 5.00 M Na2S2O3. The stepwise formation equilibria are
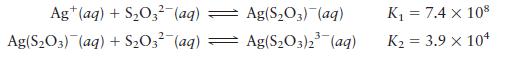
Transcribed Image Text:
Ag+ (aq) + SO3 (aq) Ag(S03) (aq) + SO3- (aq) Ag(S03) (aq) Ag(SO3)2(aq) K = 7.4 x 108 K = 3.9 x 10
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The concentrations of the ligand and metal ion in the mixed solu tion before any reaction occurs are ...View the full answer

Answered By

Saud Ur Rehman
Evaluating manufacturing processes by designing and conducting research programs; applying knowledge of product design, fabrication, assembly, tooling, and materials; conferring with equipment vendors; soliciting observations from operators. Developing manufacturing processes by studying product requirements; researching, designing, modifying, and testing manufacturing methods and equipment; conferring with equipment vendors. Keeping equipment operational by coordinating maintenance and repair services; following manufacturer's instructions and established procedures; requesting special service.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the equilibrium concentrations of NH 3 , Cu 2+ , Cu(NH 3 ) 2+ , Cu(NH 3 ) 2 2+ , Cu(NH 3 ) 3 2+ , and Cu(NH 3 ) 4 2+ in a solution prepared by mixing 500.0 mL of 3.00 M NH 3 with 500.0 mL...
-
The solubility-product constant for Ce(IO3)3 is 3.2 x 10-10. What is the Ce3+ concentration in a solution prepared by mixing 50.00 mL of 0.0450 M Ce3+ with 50.00 mL of? (a) Water? (b) 0.0450 M IO3-?...
-
Calculate [HY3-] in a solution prepared by mixing 10.00 mL of 0.010 0 M VOSO4, 9.90 mL of 0.010 0 M EDTA, and 10.0 mL of buffer with a pH of 4.00.
-
According to Electronic Designs 2012 Engineering Salary Survey, the mean base salary of a software engineering manager is $126,417the highest mean among all types of engineers. In contrast, a...
-
A photon is scattered at an angle 0 = 120 by a stationary free electron. As a result, the electron acquires a kinetic energy T = 0.45 MeV. Find the energy that the photon had prior to scattering.
-
Why do optical signals used in fiber optic cables have a very short wave length?
-
Respondents Varsity Brands, Inc., Varsity Spirit Corporation, and Varsity Spirit Fashions & Supplies, Inc., design, make, and sell cheerleading uniforms. Respondents have obtained or acquired more...
-
Carey Company had sales in 2016 of $1,560,000 on 60,000 units. Variable costs totaled $900,000, and fixed costs totaled $500,000. A new raw material is available that will decrease the variable costs...
-
The figure depicts Jack - in - the - box: "Jack" is attached inside a box by a spring, as shown. You estimate Jack's mass to be 0 . 4 k g . As so often happens, a sign tells you the spring constant:...
-
You have two salts \(\mathrm{AgX}\) and \(\mathrm{AgY}\) with very similar \(K_{\mathrm{sp}}\) values. You know that the \(K_{\mathrm{a}}\) value for \(\mathrm{HX}\) is much greater than the...
-
A solution contains 1.0 10 4 M Cu + and 2.0 10 3 M Pb 2+ . If a source of I is added to this solution gradually, will PbI 2 (K sp = 1.4 10 8 ) or CuI (K sp = 5.3 10 12 ) precipitate first?...
-
Two equal and opposite charges are attached to the ends of a rod of length s. The rod rotates counterclockwise in the x-y plane with angular speed = ck. The electric dipole moment of the system at t...
-
Calculate the present value as at 1 January 2020 of the following payment: .a single payment of euros 2,000 payable on 1st july 2024 .a single payment ofeuros 5000 payable on 31st dec 2031 assume...
-
Ethics is the process of making decisions which reflect moral goodness. We all want strategic decisions that reflect moral goodnessthey quite literally make us feel better. Strategy involves a...
-
Clare is saving up money to buy a car. Clare puts $9,635.00 into an account which earns 10% interest, compounded monthly. How much will she have in the account after 10 years? 2 answers
-
Find out how much a retirement account based on a principal of $100509 compounded 5% quarterly after 29 years is worth? Round your answer to 2 decimal places 2 answers
-
A ramp is being built to access a door that is 1.5 feet above the ground. Find the length of the ramp if the angle of elevation must be 8 degrees.
-
Did the Hyatts accept the list of contract terms included in the computer box? Why or why not? What is the name used for this type of e-contract? Ted and Betty Hyatt live in California, a state that...
-
Les has collected stamps in his spare time for years. He purchased many of his stamps at a price much lower than the current market value. Les recently lost his job as a carpenter. Since his wife...
-
Calculate the density of each of the following metals from the data given: (a) Aluminum, fcc structure, atomic radius 143 pm; (b) Potassium, bcc structure, atomic radius 227 pm.
-
Calculate the pressure exerted by 1.00 mol H 2 S, behaving as (a) An ideal gas; (b) A van der Waals gas when it is confined under the following conditions: (i) At 273.15 K in 22.414 L; (ii) At 800. K...
-
Suppose that 0.473 g of an unknown gas that occupies 200. mL at 1.81 atm and 25C was analyzed and found to contain 0.414 g of nitrogen and 0.0591 g of hydrogen. (a) What is the molecular formula of...
-
2 Consider the market for X. Suppose it is competitive. Two types of firms produce X. Type 1 has a MC function given by MC = (Q-10) + 100. Type 2 has a MC function given by MC2 = (Q2-8) + 100. Fifty...
-
1. What role does Symbolic Interaction Theory and Conflict Theory play in technology? 2. Include the perspective of both theories, whether it's positive or negative.
-
A rural village has one family physician. The nearest hospital, 25 miles away, is a thirty-bed facility owned by the physicians who practice in the rural county. Explain why village residents are...

Study smarter with the SolutionInn App


