Complete a Lewis structure for the compound shown below, then answer the following questions. How many carbon
Question:
Complete a Lewis structure for the compound shown below, then answer the following questions. How many carbon atoms are sp2 hybridized? How many CON bonds are formed by the overlap of an sp3 hybridized carbon with an sp3 hybridized nitrogen? How many lone pairs of electrons are in the Lewis structure of your molecule? How many π bonds are present?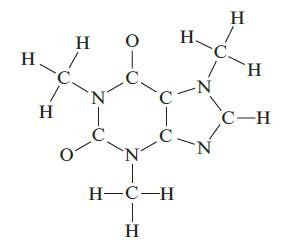
Transcribed Image Text:
H H H N T O 0- C N H-C-H T H H. C. -C -N N H H C-H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Answer There are four carbon atoms sp2 hybridized There are three CON bonds formed by ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many carbon atoms of citronellal would be radioactively labeled if the acetic acid used in the experiment were enriched with 14C at C-1 instead of at C-2? Identify these carbon atoms.
-
How many carbon atoms of citronellal would be radioactively labeled if the acetic acid used in the experiment were enriched with 14C at C-1 instead of at C-2? Identify these carbon atoms.
-
Complete the Lewis structure of the compound shown below and indicate which of the following statements are true. The C-N bond is formed from overlap of an sp 3 hybrid orbital from the carbon atom...
-
Using Figure 7. 4, show how the level of oil imports and the price level would be affected if the country represented in that figure acted to internalize national security issues, but ignored climate...
-
The time spent (in days) waiting for a kidney transplant for people ages 3549 can be approximated by a normal distribution, as shown in the figure. (a) What waiting time represents the 80th...
-
Write a method called surroundWith that takes an integer x and an integer y as parameters and surrounds all nodes in the list containing the value x with new nodes containing the value y. In...
-
The payroll disbursements were for two persons named Ciotty and Ciotti with the same first name and address. The interesting observation is that Ciotty is dated February 28, 2019, or after while...
-
The post-closing trial balance for Horner Co. is shown below. The subsidiary ledgers contain the following information: (1) accounts receivableB. Hannigan $2,500, I. Kirk $7,500, and T. Hodges...
-
Let 1 f(z) = (2-1)(x-2) Find the Laurent expansion of f about z = 0 in the region {ze C: 1
-
Ming Chen started a business and had the following transactions in June. Create the following table similar to Exhibit 1.9 and use additions and subtractions to show the dollar effects of the...
-
In which of the following diatomic molecules would the bond strength be expected to weaken as an electron is removed? a. H 2 b. B 2 c. C 2 2- d. OF
-
Ronald Robinson, Wyman Robinson, and Friendly Discount Auto Sales (appellants) appeal from the granting of summary judgment in favor of appellee Mike Durham (Durham). The facts material to this...
-
HMK Enterprises would like to raise $12 million to invest in capital expenditures. The company plans to issue five-year bonds with a face value of $1000 and a coupon rate of 5.1% (annual payments)....
-
You may not use the knn or dist commands in R for this question. You may perform calculations by hand (and attach to assignment) or manually in R (including commands and output in the document)....
-
Suppose that both Mexico and the U.S. had roughly equal GDP per capita income in the year 1800 at $1,000 apiece. Over the course of 100 years Mexicos GDP per capita growth rate was 2% while the U.S....
-
Suppose $3,200 CAD could be used to purchase $1, 828.57 USD last year. Part (a): What was the nominal exchange rate last year? Be sure to quote your answer in the proper format (example: CAD 1 USD)....
-
Perry Mason asked his secretary Della Street to take her car and deliver a pleading to the courthouse for filing. Driving to the courthouse, Della accidentally ran a stop sign and caused a driver to...
-
Objective: Students will apply concepts of Java Generics and Red-Black Trees in this programming assignment. Assignment Description: In this class, we have learned a couple of advanced data...
-
The data in Silver-Q represent the price in London for an ounce of silver (in U.S. $) at the end of each quarter from 2004 through 2009. a. Plot the data. b. Develop an exponential trend forecasting...
-
Evaluate the function at the given value(s) of the independent variable. Simplify the results. (x) = cos 2x (a) (0) (b) (- /4) (c) (/3) (d) ()
-
A solution of phosphoric acid was made by dissolving 10.0 g H 3 PO 4 in 100.0 mL water. The resulting volume was 104 mL. Calculate the density, mole fraction, molarity, and molality of the solution....
-
What is ion pairing?
-
Common commercial acids and bases are aqueous solutions with the following properties: Calculate the molarity, molality, and mole fraction of each of the preceding reagents. Hydrochloric acid Nitric...
-
Shawn has reached retirement. To supplement his pension income, he has accumulated $500,000 in non-registered investments. He wants to receive regular payments from this amount. He would like to...
-
Why might it be important to consider tone and communication style- including word choices in our engagement with others esp. family and those we need to connect with in the organization?
-
David and Lucy were married. When Lucy died last September, she owned: A Registered Retirement Savings Plan (RRSP) worth $375,000; A Tax-Free Savings Account (TFSA) worth $40,000; and A...

Study smarter with the SolutionInn App


