Complete and balance each of the following equations: (a) FeS (s) + HCl(aq) (b) H(g) +
Question:
Complete and balance each of the following equations: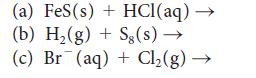
Transcribed Image Text:
(a) FeS (s) + HCl(aq) → (b) H₂(g) + Sg(s) → (c) Br(aq) + Cl₂(g) →
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Completed and balanc...View the full answer

Answered By

SHIVAM SHUKLA
I TEACH STUDENTS IN MY HOME FROM THE LAST THREE YEARS
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Complete and balance each of the following molecular equations (in aqueous solution); include phase labels. Then, for each, write the net ionic equation. a. NaOH + HNO3 b. HCl + Ba(OH)2 c. HC2H3O2...
-
Complete and balance each of the following molecular equations (in aqueous solution); include phase labels. Then, for each, write the net ionic equation. a. Al(OH)3 + HCl b. HBr + Sr(OH)2 c....
-
Complete and balance each of the following molecular equations, including phase labels, if a reaction occurs. Then write the net ionic equation. If no reaction occurs, write NR after the arrow. a....
-
Which sensing structure helps a company test and make sense of new ideas generated through other types of sensing? Social listening Crowdsourcing Blue ocean Pilot test
-
Does the following manifesto make sense? Explain briefly. Were a darn successful company. Our book rate of return has exceeded 20 percent for five years running. Were determined that new capital...
-
Marietta Wood Products (MWP) manufactures disposable chopsticks for the restaurant industry at its highly automated production facility in China. The main raw material used to produce the chopsticks...
-
Consider an asset whose price follows the geometric Brownian motion process where \(z\) is a standard Wiener process. (a) At time \(t\) (when \(S(t)\) is known), what is the expected value of the...
-
Financial statements for Allendale Company follow: ALLENDALE COMPANY Balance Sheets As of December 31 2019 2018 Assets Current assets Cash $ 40,000 $ 36,000 Marketable securities 20,000 6,000...
-
Please use CX programmer to demonstrate the answer Image transcription text LABWORK 3 PLC 3 - Conveyor & Puncher OBJECTIVES: programing. Design of a WAIT state. Pulse generation function, TOPICS:...
-
Complete and balance the following equations: (a) AlO3(s) + OH (aq) (b) AlO3(s) + H3O+(aq) + HO(1) (c) B(s) + NH3(g)
-
(a) Nitrous acid reacts with hydrazine in acidic solution to form hydrazoic acid, HN 3 . Write the chemical equation and determine the mass of hydrazoic acid that can be produced from 15.0 g of...
-
In Exercises 1 through 20, compute all first-order partial derivatives of the given function. f(x, y) = xe x+2y
-
During the year, the company had the following data Beginning inven Ending Finished Direct materials Direct labour per Variable manufa Fixed manufactu Variable selling Fixed selling cos REQUIRED (USE...
-
Consider a unimolecular reversible, elementary step reaction of the type: Equilibrium constant = K AR Given M = CRO/CAO and the conversion of A at equilibrium is fae, find the conversion of A as a...
-
I hope these notes will help you in writing your close reading papers due next week. Close Reading 1. Analysis in very fine detail, zoomed in, and preoccupied with the tiniest mechanical efforts of...
-
City Beverages Company provides a complete product line of beer, soft drink, and juice products. The following table shows the prices and quantities of the three products from the year of 2020 to...
-
(Sovereign Default Model) Let it be a risk free rate on the T-Bills. Let D, be the amount of debt. When the government issues the amount of debt Dt and defaults on it, the household consumption with...
-
The best management accounting system provides managers with all the information they would like to have. Do you agree with this statement? Why or why not?
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
The molecular electrostatic potential maps for LiH and HF are shown here. Does the apparent size of the hydrogen atom (shown as a white sphere) tell you whether it is an electron acceptor or an...
-
For H + 2 , explain why H aa is the total energy of an undisturbed hydrogen atom separated from a bare proton by the distance R.
-
Distinguish between the following concepts used to describe chemical bond formation: basis set, minimal basis set, atomic orbital, molecular orbital, and molecular wave function.
-
Provide a PESTLE analysis of the United Kingdom with particular depth given to its culture for the online food industry and how it's growing?
-
Given the different entries and thesis statement, write a five-point outline for the text that follows. Observe the standard format. Due to the mounting issues and controversies concerning global...
-
In a Lean-Agile Center of Excellence (LACE) an argument began between Lee, the Director of Software, and Clair, the Vice President of Human Resources. Clair wants to launch an Efficiency Agile...

Study smarter with the SolutionInn App


