Complete and balance the following equations: (a) AlO3(s) + OH (aq) (b) AlO3(s) + H3O+(aq) +
Question:
Complete and balance the following equations: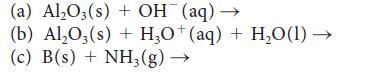
Transcribed Image Text:
(a) Al₂O3(s) + OH¯ (aq) → (b) Al₂O3(s) + H3O+(aq) + H₂O(1) → (c) B(s) + NH3(g) →
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
complete and balance ...View the full answer

Answered By

Sheetal Singhal
I am currently working on data science and machine learning project and have high command in python, java, javascript, html
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Complete and balance the following nuclear equations by supplying the missing particle: 98 53 59 0
-
Complete and balance the following nuclear equations by supplying the missing particle: ISK +-Ye (orbital electron)-? SiH 14 235
-
Complete and balance the nuclear equations for the following fission reactions: 235 + 239 94 144
-
Problem 5-47 Amortizing Loans and Inflation (LO3) Suppose you take out a $108,000, 20-year mortgage loan to buy a condo. The interest rate on the loan is 5%. To keep things simple, we will assume you...
-
Respond to the following comments: a. I like the IRR rule. I can use it to rank projects without having to specify a discount rate. b. I like the payback rule. As long as the minimum payback period...
-
After substantial marketing research, Taoyuan Corporation management believes that it can make and sell a new battery with a prolonged life for laptop computers. Management expects the market demand...
-
An alternative to using \(d=1 / u\) in a binomial model is to use the available degree of freedom by setting \(p=1 / 2\). (a) Let \(p=1 / 2\), and find the values of \(u\) and \(d\) that satisfy the...
-
You are given the following information about We-Save-Um Animal Rescue (WSU): Number of dogs rescued and placed by WSU600 Average length of stay for a dog10 days Daily cost of feeding one dog$. 90...
-
The following information is for Kon Inc. for the year ended December 31, 2021: Common shares, Jan. 1 $20,000 Common shares issued during year 10,000 Retained earnings, Jan. 1 58,000 Office expense...
-
Identify the oxidation number of tin in the following compounds and ions: (a) Sn(OH) 6 2 ; (b) SnHPO 3 ; (c) NaSn 2 F 5 .
-
Complete and balance each of the following equations: (a) FeS (s) + HCl(aq) (b) H(g) + Sg(s) (c) Br(aq) + Cl(g)
-
Use the Stenback Media Corporation data in Short Exercise S14-7 and the results you calculated from the requirements. Prepare Stenbeck Medias statement of cash flows indirect method for the year...
-
You plan to deposit amounts as shown at the beginning of this year, and at the beginning of every year until five year from now, into a bank which will give you annual interest. The following table...
-
For the question, please fill in the box that says "(???)".? The Question contains 2 parts because the question was too long to capture in a single screen ? Date 1 July 2019 1 June 2020 30 June 2020...
-
Go to the "Monthly Rounds and Revenue" tab. Management wants to know the number of rounds of golf played each month based on the "Participation Category" (Column D) and the monthly rounds of golf...
-
Corporate Stationery and Furniture Supplies Pty Ltd was acquired and registered as a company on 1 July 2021 under the rebranded name as listed. The company is classified as a Proprietary Limited (Pty...
-
CURRENT MONTHLY LIVING EXPERPSESde a breakdown of the total amount listed in the case study -use your discretion) Food/housekeeping Insurance (e.g. motor vehicles, home contents/ building, medical,...
-
Fixed costs are really variable. The more you produce, the smaller the unit cost of production. Is this statement correct? Why or why not?
-
4. Jobe dy -Y 2 et by
-
The first ionization potential of ground-state He is 24.6 eV. The wavelength of light associated with the 1s2 p 1 P term is 58.44 nm. What is the ionization energy of the He atom in this excited...
-
Give examples of AOs for which the overlap reaches its maximum value only as the internuclear separation approaches zero in a diatomic molecule. Also give examples of AOs for which the overlap goes...
-
Why is it reasonable to approximate H 11 and H 22 by the appropriate ionization energy of the corresponding neutral atom?
-
Alcohol A contains only carbon, hydrogen and nitrogen. 5.00 g alcohol A undergo complete combustion in oxygen to form 12.9 g CO2 and 2.65 g HO, releasing a huge amount of heat. Alcohol A (I) + O2 (g)...
-
(c) Given that the Kap of Mg(OH)2 is 5.6 x 1012. Calculate the pH of saturated Mg(OH)2 solution. (4 marks) (d) Given that the pK, of acetic acid is 4.76 and the pK, ethylamine (CH3CH2NH2) is 3.19....
-
In the use of credit cards to purchase an item, the credit card company now has information about the owner. Does the credit card company have the right to sell information (name, address, and phone...

Study smarter with the SolutionInn App


