Consider the ammonia synthesis reaction where G =33.3 kJ per mole of N 2 consumed at 25C.
Question:
Consider the ammonia synthesis reaction
![]()
where ΔG =–33.3 kJ per mole of N2 consumed at 25°C. For each of the
following mixtures of reactants and products at 25°C, predict the direction in
which the system will shift to reach equilibrium.
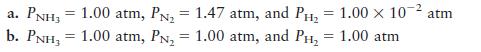
Transcribed Image Text:
N(g) + 3H(g) = 2NH3(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a We can predict the direction of the shift to equilibrium by calculating the value of AG ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the reaction 2NO2(g) N2O4(g) For each of the following mixtures of reactants and products at 25oC, predict the direction in which the reaction will shift to reach equilibrium. Use...
-
For the synthesis of ammonia at 500C, the equilibrium constant is 6.0 10 2 L 2 /mol 2 . Predict the direction in which the system will shift to reach equilibrium in each of the following cases. -3 a....
-
By applying modern technology to agriculture, the United States has become the most productive food-producing nation in the world. The secret to solving the world food security problem lies in...
-
A particle moves in a medium under the influence of a retarding force equal to mk(v3 + a2v), where k and a are constants. Show that for any value of the initial speed the particle will never move a...
-
Figure 3.33 shows a manometer being used to indicate the difference in pressure between two points in a pipe. Calculate (p A - p B ). Oil (sg = 0.90) 3 ft 2'ft 6 ft Water
-
Petitioner Salman was indicted for federal securities-fraud crimes for trading on inside information he received from a friend and relative-by-marriage, Michael Kara, who, in turn, had received the...
-
Matthias Medical manufactures hospital beds and other institutional furniture. The companys comparative balance sheet and income statement for 2012 and 2013 follow. Required Calculate the following...
-
Explain entity integrity and referential integrity rules in relational model. Show how these are realized in SQL.
-
The overall reaction for the corrosion (rusting) of iron by oxygen is Using the following data, calculate the equilibrium constant for this reaction at 25C. 4Fe(s) + 30(g) 2FeO3(s)
-
One method for synthesizing methanol (CH 3 OH) involves reacting gaseous carbon monoxide and hydrogen: Calculate G at 25C for this reaction, in which carbon monoxide gas at 5.0 atm and hydrogen gas...
-
The circular disk of radius r is mounted on its shaft which is pivoted at O so that it may rotate about the vertical z 0 -axis. If the disk rolls at constant speed without slipping and makes one...
-
Now that Lucy has finished closing the hospital's financial books, she can set-up the financial books in the new accounting system. 2. Initial Post: Create a new thread and answer all three parts of...
-
What should be done, if anything, to fix the impact on ad sales to creating privacy barriers on user information? What can be done to ensure advertisers that their ad dollars will reach their target...
-
1. List the number of groups to which you now belong. Include both voluntary and involuntary. 2. How do the groups you listed affect the way you think, feel, and act in your society? 3. What is your...
-
Ratio analysis is often used as a starting point for potential investors. Explain the key strengths and weaknesses of ratio analysis. Why are these strengths and weaknesses (provide two of each)?...
-
In what way does this Ted Talk describe the production process? In a similar fashion to what that AJ Jacobs did, describe how the production process has brought value to your life. In other words,...
-
Refer to the data in Exercise 6-31. Draw the cost flow diagram assuming that Tiger Furnishings uses direct labor costs to allocate overhead costs.
-
Determine whether the lines are parallel, perpendicular, or neither. 2x + 3y = -12, 2y - 3x = 8
-
The molar concentration of HCl in hydrochloric acid is reduced to 12% of its initial value by dilution. What is the difference in the pH values of the two solutions?
-
Salts are often used to create solutions with specific pH values. Suppose you need to prepare a salt solution with a pH of about 4.5 and have available sodium dihydrogen phosphate, NaH 2 PO 4 , and...
-
Calculate the standard reaction Gibbs free energy for the following cell reactions: 4+ (a) 2 Ce (aq) + 3I (aq) 2 Ce(aq) + 13 (aq), Ecell +1.08 V (b) 6 Fe+(aq) +2 Cr+(aq) + 7 HO(1) 6 Fe+ (aq) + CrO2...
-
Tasks 4: Matlab code for predicting the average time Goal: Develop a Matlab code that is able to predict the average time that your robot will be able to complete a randomly created maze. You must...
-
3. (a) The general formula of the structure factor for a cubic crystal can be written as: Fhkl fmcos (2(hxm + kym + lzm)) = (iv) Define all the terms used in equation (1) above. (v) (1) (4 marks)...
-
IV. Disturbance Accommodating Controller (DAC): Use the same (A,B,C) as in Problem II. (x=. =Ax+ Bu+Tup Let [up=0zD with Disturbance Generator &r= y=Cx ZD=FZD = a) Let u, sint+, cost (sint & cost)....

Study smarter with the SolutionInn App


