Draw the Lewis structure or symbol of each reactant, identify the Lewis acid and the Lewis base,
Question:
Draw the Lewis structure or symbol of each reactant, identify the Lewis acid and the Lewis base, and then draw the Lewis structure of the product (a complex) for the following Lewis acid–base reactions: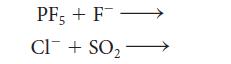
Transcribed Image Text:
PF, + F- CI+SO₂1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a b FF Lewis acid ...View the full answer

Answered By

Benish Ahmad
I'm a professional software engineer. I'm lectutrer at GCUF and I have 3 years of teaching experience. I'm looking forward to getting mostly computer science work including:
Programming fundamentals
Object oriented programming
Data structures
object oriented design and analysis
Database system
Computer networks
Discrete mathematics
Web application
I am expert in different computer languages such as C++, java, JavaScript, Sql, CSS, Python and C#. I'm also have excellent knowledge of essay writing and research. I have worked in other Freelancing website such as Fiverr and Upwork. Now I have finally decided to join the SolutionInn platform to continue with my explicit work of helping dear clients and students to achieve their academic dreams. I deliver plagiarism free work and exceptional projects on time. I am capable of working under high pressure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Draw the Lewis structure or symbol of each reactant, identify the Lewis acid and the Lewis base, and then draw the Lewis structure of the product (a complex) for the following Lewis acidbase...
-
Draw the Lewis structure or symbol for each of the following species and identify each one as a Lewis acid or Lewis base: (a) NH3; (b) BF3; (c) Ag; (d) F; (e) H.
-
Draw the Lewis structure or symbol for each of the following species and identify each one as a Lewis acid or Lewis base: (a) SO; (b) I; (c) CH3S (the C atom is the central atom); (d) NH; (e) NO.
-
Pritchett Company reported the following year-end data: Cash $ 25,000 8,000 Short-term investments Accounts receivable (current) Inventory 19,500 27,500 Prepaid (current) assets 11,000 Total current...
-
Non-financial measures of quality and time. Worldwide Cell Phones (WCP) has developed a cell phone that can be used anywhere in the world (even countries like Japan that have a relatively unique cell...
-
Amber Mining and Milling, Inc., contracted with Truax Corporation to have constructed a custom-made lathe. The machine was completed and ready for use on January 1, 2011. Amber paid for the lathe by...
-
Explain the role of a questionnaire in the research process. What should be the role of the client during the questionnaire development process?
-
1. What advantages does a subscription pricing model offer a business? 2. Notice that several of Bark & Co.s idea for new businesses have failed. Is this unusual? Why is it important for businesses...
-
Suppose the price of gasoline increases by 20% and the demand for gasoline decreases by 10%. What is the price elasticity of demand of gasoline? Answer: x
-
The structure below shows a hydrated d-metal ion. Draw the structure of the conjugate base of this complex.
-
For the exercise in this topic, base your answers on the potentials listed in Table 6M.1 or Appendix 2B, with the exception of the reduction and oxidation of water at pH = 7: Three electrolytic cells...
-
Review the Decision Ethics case from the first part of this chapter involving the cashier. The guidance answer suggests that you should not comply with the assistant managers request. Required...
-
It was not clear that the revenue in the income statement needed to be reported net of the district-paid expenses for those cases in which Edison was not the primary ____________.
-
You want to know what team members are available immediately for a quick video conference. They are all workaholics and glued to their mobile devices.
-
According to the SEC, Adelphia purchased land from members of the Rigas family in terms of a contract in which a clause stated that the ____________ rights to the land would revert back to the owner...
-
What will be H&Ms cost of equity if the equity market risk premium is 6 percent?
-
Under the competitive equilibrium assumption, the terminal value in the discounted abnormal profit growth model is the present value of abnormal profit in the terminal year times minus one,...
-
Operating budgets for the DiPaolo Company reveal the following information: net sales, $450,000; beginning materials inventory, $23,000; materials purchased, $185,000; beginning work in process...
-
In a large midwestern university, 30% of the students live in apartments. If 200 students are randomly selected, find the probability that the number of them living in apartments will be between 55...
-
Predict the product(s) obtained when each of the following compounds is treated with a mixture of nitric acid and sulfuric acid: (a) (b) (c) (d) (e) Br
-
Predict the major product obtained when each of the following compounds is treated with fuming sulfuric acid: (a) Chlorobenzene (b) Phenol (c) Benzaldehyde (d) Ortho-Nitrophenol (e) Para-Bromotoluene...
-
For each of the following groups, identify whether it is an activator or a deactivator, and determine its directing effects: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) -OMe
-
When auditors are engaged to audit financial statements prepared on a regulatory basis, they may be requested to issue their report on a specific form designed by authorities. If they do not believe...
-
Solve the system using an augmented matrix. Show all work. x-3y=-17 2x - 6y=-26
-
Topic: Visit the Statistics Canada website and look for Canada's exports, imports, and current trade balance. How does Canada's economy benefit from being a major trading partner of the U.S.? Does...

Study smarter with the SolutionInn App


