The structure below shows a hydrated d-metal ion. Draw the structure of the conjugate base of this
Question:
The structure below shows a hydrated d-metal ion. Draw the structure of the conjugate base of this complex.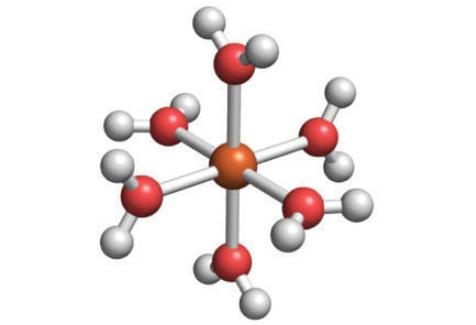
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
The image youve provided shows a metal ion in orange surrounded by water molecules which are the lig...View the full answer

Answered By

Sultan Ghulam Dastgir
The following are details of my Areas of Effectiveness English Language Proficiency, Organization Behavior , consumer Behavior and Marketing, Communication, Applied Statistics, Research Methods , Cognitive & Affective Processes, Cognitive & Affective Processes, Data Analysis in Research, Human Resources Management ,Research Project,
Social Psychology, Personality Psychology, Introduction to Applied Areas of Psychology,
Behavioral Neurosdence , Historical and Contemporary Issues in Psychology, Measurement in Psychology, experimental Psychology,
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Vasovist is the tradename of a Gd(III) complex which was the first intravascular contrast agent (see Box 4.3) approved in the EU for use in magnetic resonance angiography. Interactions between the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The Delegator Instructions: You are a manager for a busy, medium-sized company. You have recently been ill, and the doctor has told you that you need to cut your workload in half. Your...
-
5.Construct a Histogram for the frequency distribution below containing record high temperatures for each of the 50 states. Class boundaries Freq 99.5-104.5 104.5-109.5 109.5-114.5 114.5-119.5...
-
Quality improvement, relevant costs, relevant revenues. TechnoPrint manufactures and sells 20,000 high-technology printing presses each year. The variable and fixed costs of rework and repair are as...
-
On January 1, a company issued 7%, 15-year bonds with a face amount of $90 million for $82,218,585 to yield 8%. Interest is paid semiannually. What was interest expense at the effective interest rate...
-
What makes a question bad? Develop three examples of bad questions. Rewrite your examples so they could be judged as good questions.
-
OpenSeas, Inc., is evaluating the purchase of a new cruise ship. The ship would cost $500 million, but would operate for 20 years. OpenSeas expects annual cash flows from operating the ship to be $70...
-
Do the following two parts: (a) Find the sum-of-products expansion of the Boolean function F (w, x, y, z) that has the value 1 if and only if an odd number of w, x, y, and z have the value 1. (b)...
-
The autoprotolysis constant, K hw , for heavy water, D 2 O, at 25C is 1.35 * 10 15 . (a) Write the chemical equation for the autoprotolysis (more precisely, the autodeuterolysis, because a deuteron...
-
Draw the Lewis structure or symbol of each reactant, identify the Lewis acid and the Lewis base, and then draw the Lewis structure of the product (a complex) for the following Lewis acidbase...
-
Estimate G(1.0) if G obeys the differential equation dG/dt = G - 1 with initial condition G(0) = 2. Use Euler's method with t = 0.2 for five steps. Compare with the exact answer in Exercise 17.
-
You wish to use a converging mirror to form an image that is \(N\) times larger than the object. In terms of \(N\) and \(R\), the radius of curvature of the mirror, at what distance ofrom the mirror...
-
A travel visa cannot be issued until an application is completed and a recent photo is included. Revise the following statements to make them more positive and courteous.
-
We are sorry to let you know that we can offer the 30 percent rebate only to the first 25 buyers, so hurry up! Revise the following statements to make them more positive and courteous.
-
BTW, Madison blew a gasket when the manager accused the whole department of ripping off pricey office supplies. Revise the following to make the tone conversational yet professional.
-
Civil Service exams were once required for federal government jobs, but they were phased out subsequent to the passage of antidiscrimination laws. Revise the following sentences to use plain language...
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
A hard-working horse can lift a 350. lb. weight 100. ft. in one minute. Assuming the horse generates energy to accomplish this work by metabolizing glucose: C 6 H 12 O 6 (s) + 6O 2 (g) 6CO 2 (g) +...
-
Under what conditions is the distribution of products in an ideal gas reactions system at equilibrium unaffected by an increase in the pressure?
-
Identify which of the following compounds is most activated toward electrophilic aromatic substitution. Which compound is least activated? Br NO2 NO2 OMe .
-
Consider the following relations on A = {0, 1, 2, 3, 4}. Determine which are reflexive which are symmetric, which are transitive, and which are equivalence relations (Circle/State all that apply and...
-
The East Division of Abel Company reported the following information for 2025: Variable costs per unit Average operating assets Return on investment Margin ... Minimum required rate of return Sales...
-
Journal entry worksheet > A B Rec-D Record the closure of variable overhead cost variances. Note: Enter debits before credits. Event 2 General Journal Debit Credit

Study smarter with the SolutionInn App


