How many chelate rings are present in (a) [Ru(ox) 3 ] 3 ; (b) [Fe(trien)] 3+ (trien
Question:
How many chelate rings are present in
(a) [Ru(ox)3]3–;
(b) [Fe(trien)]3+ (trien is triethylenetetramine);
(c) [Cu(dien)2]2+?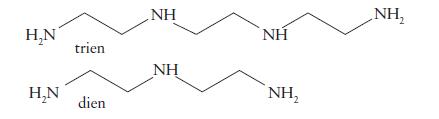
Transcribed Image Text:
H₂N H₂N triển dien NH ΝΗ ΝΗ NH₂ NH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
In summary a Ruox33 has 3 chelate rings b Fetrien3 has 1 chelate ring c Cud...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
In Problems 1968, solve each equation, if possible. 3 2 -x + 2 1 2 1 2
-
Add or subtract as indicated. 4x + 1 3x + 2 -2x - 3 3x + 2
-
How many chelate rings are present in each of the following complexes? Assume that all the donor atoms are involved in coordination. (a) [Cu(trien)] 2+ ; (b) [Fe(ox) 3 ] 3 ; (c) [Ru(bpy) 3 ] 2+ ; (d)...
-
Pepsin is the principal digestive enzyme of gastric juice. A 1.40 g sample of pepsin is dissolved in enough water to make 4.50 mL of solution. The osmotic pressure of the solution is found to be...
-
Suppose three processer, p1, p2, and p3, are attempting to use a machine with interrupt-driven I/O concurrently. Suppose p1 has tcompute = 20 and tdevice = 50, p2 has tcompute = 30 and tdevice =10,...
-
Journalize the following transactions, using the allowance method of accounting for uncollectible receivables: Feb. 12. Received $800 from Leo Jorgenson and wrote off the remainder owed of $2,400 as...
-
For each of the following, calculate the standard error of the difference \(\left(s \mathrm{X}^{-} 1-\mathrm{X}^{-} ight.\) \(2)\). a. N = 35, s = 1.50, N = 35, $2 = 3.25 b. N = 4, s = 4.30, N = 4,...
-
You are considering opening a copy service in the student union. You estimate your fixed cost at $15,000 and the variable cost of each copy sold at $.01. You expect the selling price to average $.05....
-
Aloha Inc. has 7 percent coupon bonds on the market that have 12 years left to maturity. If the YTM on these bonds is 8.1 percent, what is the current bond price?
-
Identify the type of structural isomerism that exists in each of the following pairs of compounds or ions: (a) [Pt(OH)4] [PtCl6] and [PtCl(OH)4] [PtCl4] (b) [Cr(en)3] [Co(ox)3] and [Co(en)3][Cr(ox)3]...
-
Iron in blood serum can be measured by reducing it to Fe 2+ and reacting it with ferrozine to form [Fe(ferrozine) 3 ] 4 , a purple complex that has a maximum absorbance at 562 nm. The molar...
-
One of the following statements about promissory notes is incorrect. The incorrect statement is: (a) The party making the promise to pay is called the maker. (b) The party to whom payment is to be...
-
EXCEL HELP NEEDED 1 4 5 8 9 File 10 DHE BAE 12 13 14 15 16 17 11 Savings at t = 10 32 33 course hero - Google Search X about:blank 34 35 36 37 38 39 40 Excel template HH Home Insert Draw Page Layout...
-
Maicom Construction Materials Inc. , hereinafter referred to as "MCM", is a construction materials company established in Moncton, New Brunswick. Its facilities (warehouse, store and offices) are...
-
HOMEWORK: Adjustments: Tara has the following PARTIAL Trial Balance: Account Name Interest Receivable Supplies Prepaid Rent Equipment Accumulated Depreciation Unearned Revenue Utility Payable Revenue...
-
Macmillan Learning Attempt 1 Charge q = -q is located at position (0, d). Charge q2 = -3q1 is located at position (d, 0). Charge 3 = = 3q1 is located at position (2d, 2d). d d 9 d d X Determine the...
-
Calculate the magnitude of the electric field at one corner of a square 2.12 m on a side if the other three corners are occupied by 5.25x10-6 C charges. Express your answer to three significant...
-
Give three reasons why someone would want to own and operate a restaurant.
-
A fuel pump sends gasoline from a car's fuel tank to the engine at a rate of 5.88 10-2 kg/s. The density of the gasoline is 735 kg/m3, and the radius of the fuel line is 3.18 10-3 m. What is the...
-
For an ensemble consisting of a mole of particles having two energy levels separated by 1000. cm 1 , at what temperature will the internal energy equal 3.00 kJ?
-
The three lowest energy levels for atomic carbon (C) have the following energies and degeneracies: Determine the electronic contribution to C V for atomic C at 100. K. Level (n) Energy (cm)...
-
Carbon dioxide has attracted much recent interest as a greenhouse gas. Determine the vibrational contribution to C V for CO 2 , where 1 = 2349 cm -1 , 2 = 667 cm -1 (doubly degenerate), and 3 =...
-
Required: Prepare a flexible budget performance report for February. Note: Indicate the effect of each variance by selecting "F" for favorable, "U" for unfavorable, and "None" for no effect (i.e.,...
-
After WW 2 ended, North America experienced an economic boom, allowing companies and organizations to expand their operations, thereby creating a demand for education in areas like business and...
-
Designing quality into a product has many benefits. Some of those benefits are customer satisfaction, cost savings, and building a positive reputation. Customer satisfaction is the main goal by any...

Study smarter with the SolutionInn App


