The three lowest energy levels for atomic carbon (C) have the following energies and degeneracies: Determine the
Question:
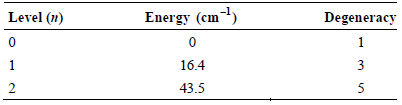
Determine the electronic contribution to CV for atomic C at 100. K.
Transcribed Image Text:
Level (n) Energy (cm) Degeneracy 1 1 16.4 3 43.5 3. 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
Beginning the expression for internal energy derived in ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The lowest four energy levels for atomic vanadium (V) have the following energies and degeneracies: What is the contribution to the average energy from electronic degrees of freedom for V when T =...
-
Calculate the three lowest energy levels, together with their degeneracies, for the following systems (assume equal mass distinguishable particles): a. Three non-interacting spin particles in a box...
-
The molar heat capacities for carbon dioxide at 298.0 K are Cv = 28.95 J K-1 mol-1 Cp = 37.27 J K-1mol-1 The molar entropy of carbon dioxide gas at 298.0 K and 1.000 atm is 213.64 JK-1mol-1. a....
-
Mrs Anh Thuy is a 43 year old lady admitted following an incidence of blurred vision, numbness down the right side and a sharp pain in her head. A neighbour found her on the ground unable to move or...
-
(a) state the domain of the function, (b) identify all intercepts, (c) find any vertical or slant asymptotes, and (d) plot additional solution points as needed to sketch the graph of the rational...
-
A continuous, stirred tank reactor is initially filled with solvent. At \(t=0\), reactant, A, is fed into the tank at a rate of \(\dot{v}_{a o}\left(\mathrm{~m}^{3} / \mathrm{min} ight)\). The...
-
Can earned value be used to manage a portfolio of projects? If so, how would this analysis proceed?
-
All-Star Bat Manufacturing, Inc., supplies baseball bats to major and minor league baseball teams. After an initial order in January, demand over the six-month baseball season is approximately...
-
Five Defendants are on trial for burglarizing a cash vault at a casino on the Las Vegas Strip. The prosecution alleges that the Defendants hacked the security cameras, wore SWAT clothing, and used...
-
X Ltd. has 10 lakhs equity shares outstanding at the beginning of the accounting year 2016. The appropriate P/E ratio for the industry in which D Ltd. is 8.35. The earnings per share is Rs. 15 in the...
-
For an ensemble consisting of a mole of particles having two energy levels separated by 1000. cm 1 , at what temperature will the internal energy equal 3.00 kJ?
-
Carbon dioxide has attracted much recent interest as a greenhouse gas. Determine the vibrational contribution to C V for CO 2 , where 1 = 2349 cm -1 , 2 = 667 cm -1 (doubly degenerate), and 3 =...
-
Determine the reactions at the supports, then draw the moment diagram for each member. EI is constant. 10 k -8 ft -8 ft- 10 ft B.
-
In January 2023, Marty's Fine Pens, a business carried on as a sole proprietorship, sells a limited-edition fountain pen for $125,000. The cost of the pen is $63,000. There is a down payment of...
-
Trevor's Tool Shop is considering investing in a new machine. The company currently has $500,000 per year in sales. The company has $265,000 per year in net income. If the company invests in the new...
-
Bunga Raya Kuning Berhad is a Malaysian-based MNC that obtains 12 percent of its supplies from the U.K. manufacturers. Sixty-five percent of its revenues are from due to exports to U.K. where its...
-
American X wishes to borrow U.S. dollars at a fixed rate of interest. Corporation Y wishes to borrow Japanese Yen at a fixed rate of interest. The amounts required by the two companies are the same...
-
What is the role of education in promoting social mobility and reducing inequality, and how can educational systems be reformed to better serve diverse populations while preparing individuals for the...
-
When gaining an understanding of the client, the auditor will identify the geographic location of the client because: (a) More spread-out clients are harder to control. (b) The auditor will need to...
-
The unadjusted trial balance of Secretarial Services is as follows: SECRETARIAL SERVICES Unadjusted Trial Balance as at 31 December 2017 Account Debit Credit Cash at bank Office supplies Prepaid...
-
Compound A is an alkene that was treated with ozone (followed by DMS) to yield only 4-heptanone. Identify the major product that is expected when compound A is treated with MCPBA followed by aqueous...
-
Identify the reagents necessary to achieve each of the following transformations: Br Br - " Br Br "Br
-
Determine the configuration for every chirality center in each of the following compounds. a. b. c. - - - - - - CH- - CH- II
-
A pitcher throws a 0.60 kg ball of clay at a 4.0 kg block of wood. The clay sticks to the wood on impact, and their joint velocity afterward is 2.5 m/s. What was the original speed of the clay (in...
-
A helium-filled balloon, whose envelope has a mass of 0.29 kg, is tied to a 3.8-m long, 0.054-kg string. The balloon is spherical with a radius of 0.40 m. When released, it lifts a length h of the...
-
Give a summary of concepts and the applications for the following quistion: When a 200.0-g mass is attached to a spring, it stretches the spring by 7.50 cm. With that mass-spring system in...

Study smarter with the SolutionInn App


