Identify (a) The functional groups and (b) The chiral carbon atoms in the histidine molecule shown here.
Question:
Identify
(a) The functional groups and
(b) The chiral carbon atoms in the histidine molecule shown here.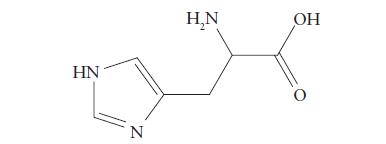
Transcribed Image Text:
HN N H,N HO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The structure youve provided is that of the amino acid histidine which is one of the twenty standard ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Identify (a) The functional groups and (b) The chiral carbon atoms in the mannose molecule shown here. H T ----- HO HO
-
(A) Identify the chiral carbon atoms in the molecule shown in the diagram on the right. (B) How many chiral atoms are there in 1,1,3-trimethylcyclohexane?
-
Micro Industries is investing $6500 in a new coffee vending machine, and expects the following cash flows over the next 4 years - Year 1 = $800, Year 2; $1400 Year 3: $2100; Year 4 $2400, Year 5...
-
Lang Enterprises was started when it acquired $4,000 cash from creditors and $6,000 from owners. The company immediately purchased land that cost $9,000. Required a. Record the events under an...
-
Find Vo using Norton's theorem for the circuit in figure. 4/0v(+ -j1n V, +2V, 10 V.
-
A stockbroker calls you and suggests that you invest in the Lauren Computer Company. After analyzing the firms annual report and other material, you believe that the distribution of expected rates of...
-
What is the function of an administrative agency?
-
What is the time value of money? Why should accountants have an understanding of compound interest, annuities, and present value concepts?
-
Consider a 10 year bond with face value $1,000, pays 6% coupon annually and has a yield-to-maturity of 7%. How much would the approximate percentage change in the price of bond if interest rate in...
-
Which of the following molecules or ions may function as a nucleophile in a nucleophilic substitution reaction: (a) OH ; (b) NH 4 + ; (c) NH 2 2; (d) H 2 O?
-
A branched hydrocarbon C 4 H 10 reacts with chlorine in the presence of light to give two branched structural isomers with the formula C 4 H 9 Cl. Write the structural formulas of (a) The...
-
Which of the following statements retrieves data from the Friends database and stores it in the FriendsDataSet? a. Me.FriendsDataSet.Fill(Friends.accdb) b....
-
You are considering buying an oven for your bakery. The oven allows you to "bake fresh cakes to order." However, baking in the store is more expensive than receiving from suppliers, and you estimate...
-
Theory and Fundamentals of Radar Operations. Explain the history of radar and the importance it has within the ATC system. How has it evolved to the equipment used by today's air traffic controllers?...
-
One of the stores of the Grocery Sales Organization (GSO) has asked for your help to try to figure out if they should hire more cashiers for the upcoming months or if they will have enough to meet...
-
The 15 questions below represent content covered from the last 5 modules. I will be randomly selecting 1 question from each module (5 questions in total) for your quiz Monday, March 20th, 2023. You...
-
The COVID-19 pandemic was a force that changed the world. Reflect on this external force when answering the questions below. Identify an organization that initiated a project in response to...
-
Because of the huge fixed cost of running pipes to everyones home, natural gas is a natural monopoly. Suppose demand is Q = 100 P and marginal revenue is MR = 100 2Q. Suppose marginal cost is $20,...
-
Suppose the index goes to 18 percent in year 5. What is the effective cost of the unrestricted ARM?
-
The same product is obtained when either d-allose or l-allose is treated with sodium borohydride in the presence of water. Explain this observation.
-
Of the eight d-aldohexoses, only two of them form optically inactive alditols when treated with sodium borohydride in the presence of water. Identify these two aldohexoses and explain why their...
-
Determine whether each of the following compounds is a reducing sugar: (a) (b) (c) CH,OH - CH, H- .
-
Oriole Corporation purchased equipment very late in 2023. Based on generous capital cost allowance rates provided in the Income Tax Act, Oriole claimed CCA on its 2023 tax return but did not record...
-
Callisto Company manufactures chemical additives for industrial applications. As the new cost accountant for Callisto, you have been assigned the task of completing the production cost report for the...
-
Tartufo Corp. entered into a 5-year lease agreement with Gelato Inc. to lease equipment beginning on January 1, 20X5. The IBR is 9% while the rate implicit in the lease is 8%. Tartufo Corp. Is aware...

Study smarter with the SolutionInn App


