Identify the arrangement (I, II, or III; all molecules are CH 2 Cl 2 ) that should
Question:
Identify the arrangement (I, II, or III; all molecules are CH2Cl2) that should possess the strongest intermolecular attractions, and justify your selection.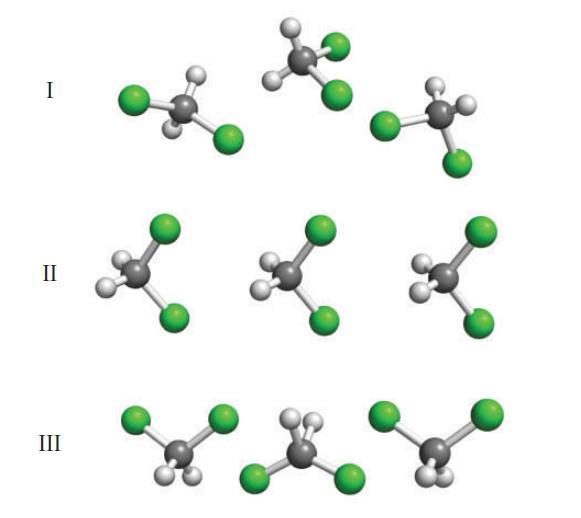
Transcribed Image Text:
H II III
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Of the three arrangementsarrangement I should have the strongest intermolecular attractionsThis is b...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Identify the arrangement (I, II, or III; all molecules are NH 3 ) that should possess the strongest intermolecular attractions, and justify your selection. I II III
-
Champion Stores are in trouble. Although the company is a major regional player with hundreds of stores in the upper Midwest, a sharp decline in the regions manufacturing economy has put the company...
-
Suppose the relationship between wage, years of education (educ), years of experience (exper), and participation in a job training program (train) is modeled as: log ( ) = + + + . Which of the...
-
???????Need answer for below questionWoolworth Ltd, After Pay Ltd, and Santos Ltd for the years 2021,2020 and 2019. Examine the consolidated Cash Flow Statements foreach companies acr f) If the firm...
-
Determine the angle ? (0? ? ? ? 180?) of the force F so that it produces a maximum moment and a minimum moment about point A. Also, what are the magnitudes of these maximum and minimum moments? 15m...
-
For the Fruity Juices scenario (Problem 13.50), formulate and solve a linear optimization model using the auxiliary variable cells method and write a short memo explaining the sensitivity...
-
Analog tape players read sound recorded on magnetic tape. The sound is recorded by magnetizing the tape longitudinally (magnetization parallel to the tape's length) with varying magnetic field...
-
This problem continues the Draper Consulting, Inc., situation from Problem 8-42 of Chapter 8. Refer to Problem 2-62 of Chapter 2. In Chapter 2, we learned that Draper Consulting, Inc., had purchased...
-
* Installing integrated software needs more memory than installing many .standalone applications True O False
-
The density of rhodium is 12.42 g cm 3 and its atomic radius is 134 pm. Is the metal close-packed or body-centered cubic?
-
(a) The van der Waals parameters for helium are a = 3.46 * 10 2 bar L 2 mol 2 and b = 2.38 * 10 2 L mol 1 . Calculate the apparent volume (in pm 3 ) and radius (in pm) of a helium atom as...
-
Ocean View Services had the following unadjusted balances at December Salaries Payable, $0; Salaries Expense, $1,400. The following transactions place at the end of 2016 and beginning of 2017: 2016...
-
Summer BBQ uses a perpetual inventory system to record inventory. All purchases and sales of merchandise inventory were on account. Company records indicate the following data for the month of June...
-
Holmes, Inc. expects net cash flow from operating activities to be $160,000, and the company plans purchases of equipment of $83,000 and repurchases of stock of $24,000. What is Holmess free cash...
-
How should you record a capital expenditure? a. Debit a liability b. Debit capital c. Debit an expense d. Debit an asset
-
Mountain Water Corp. issued common stock of $28,000 to pay off long-term notes payable of $28,000. In what section(s) would these transactions be recorded? a. Financing activities payment of note,...
-
What are the implications of divergency and complexity in the fast-food business?
-
Describe the defend strategy. List and describe the three common methods.
-
A non-charmed baryon has strangeness S = 2 and electric charge Q = 0. What are the possible values of its isospin I and of its third component I z ? What is it usually called if I = 1/2?
-
Although both the Br3- and I3- ions are known, the F3- ion does not exist. Explain.
-
The structure of TeF5 2 is Draw a complete Lewis structure for TeF5-, and explain the distortion from the ideal square pyramidal structure. (See Exercise 90.) 79. Te
-
There are two possible structures of XeF2Cl2, where Xe is the central atom. Draw them, and describe how measurements of dipole moments might be used to distinguish among them.
-
1. What are the ethical challenges Uber faces when using app-based peer-to-peer sharing technology? 2. Given that Uber uses a disruptive business model and marketing strategy, what are the risks the...
-
Choose a service environment for a company you are familiar with and list the key physical attributes of the service environment, using the three subdimensions, and discuss their impact on customer...
-
Mark Clarify the text When you feel confused while reading, where should you pause and use a strategy to understand the text? Which of these strategies help you understand the text? Look up the...

Study smarter with the SolutionInn App


