Identify the arrangement (I, II, or III; all molecules are NH 3 ) that should possess the
Question:
Identify the arrangement (I, II, or III; all molecules are NH3) that should possess the strongest intermolecular attractions, and justify your selection.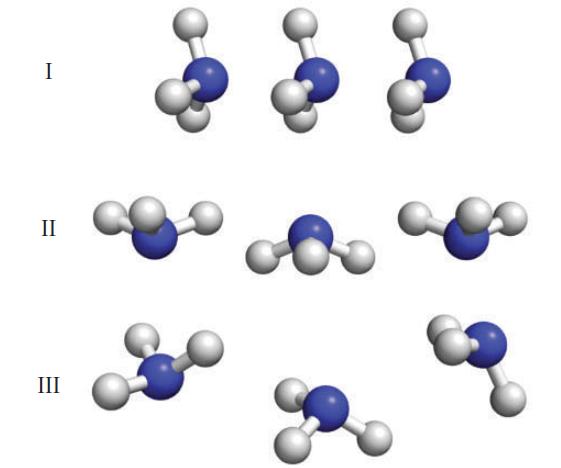
Transcribed Image Text:
I II III
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Selection I shows the weakest intermolecular attractions because all t...View the full answer

Answered By

Khurram shahzad
I am an experienced tutor and have more than 7 years’ experience in the field of tutoring. My areas of expertise are Technology, statistics tasks I also tutor in Social Sciences, Humanities, Marketing, Project Management, Geology, Earth Sciences, Life Sciences, Computer Sciences, Physics, Psychology, Law Engineering, Media Studies, IR and many others.
I have been writing blogs, Tech news article, and listicles for American and UK based websites.
4.90+
5+ Reviews
17+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Identify the arrangement (I, II, or III; all molecules are CH 2 Cl 2 ) that should possess the strongest intermolecular attractions, and justify your selection. H II III
-
Champion Stores are in trouble. Although the company is a major regional player with hundreds of stores in the upper Midwest, a sharp decline in the regions manufacturing economy has put the company...
-
Suppose the relationship between wage, years of education (educ), years of experience (exper), and participation in a job training program (train) is modeled as: log ( ) = + + + . Which of the...
-
A firm has $180 million in annual sales; $40 million of inventory and $60 million of accounts receivable. What is the inventory turnover ratio?
-
Determine the direction ? for 0? ? ? ? 180 of the force F so that it produces the maximum moment about point A. Calculate this moment. F= 400 N 2 m -3 m-
-
Use the profit model developed in Example 12.1 to implement a financial simulation model for a new product proposal and determine a distribution of profits using the discrete distributions below for...
-
The plates of a capacitor are charged using a battery, and they produce an electric field across the separation distance \(d\) between them. The two plates are now to be pushed together to a...
-
The computer workstation furniture manufacturing that Adriana Lopez started in January is progressing well. As of the end of June, Success Systems job cost sheets show the following total costs...
-
23. A convex mirror used for rear view on an automobile has a radius of curvature of 3m. If a bus is located at 5m from this mirror, find the position, nature and magnification of the image. 24. An...
-
The density of molybdenum is 10.22 g cm 3 and its atomic radius is 136 pm. Is the metal close-packed or body-centered cubic?
-
Show that the van der Waals parameter b is related to the molecular volume V mol , the volume occupied by one molecule, by b = 4N A V mol . Treat the molecules as spheres of radius r, so that V mol =...
-
Record the following transactions at (a) Gross (b) Net: 201X Sept. 3 Bought merchandise on account from Fisher Co.; terms 9/10, n/30, $8,000. Voucher no. 34 was prepared. 18 Issued check no. 481 in...
-
Michelle Darby receives cash from customers. Her other assigned job is to post the collections to customer accounts receivable. Her company has weak a. assignment of responsibilities. b. ethics. c....
-
During the year, Bernard Company had net credit sales of $45,000. At the end of the year, before adjusting entries, the balance in Accounts Receivable was $12,500 (debit) and the balance in Allowance...
-
Separation of duties is important for internal control of a. cash receipts. b. cash payments. c. Neither of the above d. Both a and b
-
Recording a dishonored note receivable McKale Corporation has a three-month, $18,000, 9% note receivable from L. Peters that was signed on June 1, 2024. Peters defaults on the loan on September 1....
-
A copy machine costs $45,000 when new and has accumulated depreciation of $44,000. Suppose Print and Photo Center discards this machine and receives nothing. What is the result of the disposal...
-
Using the data classification scheme presented in this chapter, identify and classify the information contained in your personal computer or personal digital assistant. Based on the potential for...
-
Perform the operation by first converting the numerator and denominator to scientific notation. Write the answer in scientific notation. 7,200,00/0.000009
-
Lewis structures can be used to understand why some molecules react in certain ways. Write the Lewis structures for the reactants and products in the reactions de-scribed below. a. Nitrogen dioxide...
-
The most common type of exception to the octet rule are compounds or ions with central atoms having more than eight electrons around them. PF5, SF4, ClF3, and Br3- are examples of this type of...
-
SF6, ClF5, and XeF4 are three compounds whose central atoms do not follow the octet rule. Draw Lewis structures for these compounds.
-
If you do not currently have a retirement account, what type of investment plan(s) will you use for your retirement savings (e.g., stocks, bonds, ETFs, mutual funds, or other)? Why did you choose...
-
Sarah is having difficulties with his family . Her family does not approve of her partner Tina, and they have threatened to challenge any estate planning mechanism he uses which would give Sarah...
-
Mike Fagan needs $25,000 to expand his flower shop. Find the face value of a simple discount note that will provide the $25,000 in proceeds if he plans to repay the note in 240 days and the bank...

Study smarter with the SolutionInn App


