Predict the major products of each of the following reactions and then balance the equations: (a) FeCrO4(s)
Question:
Predict the major products of each of the following reactions and then balance the equations: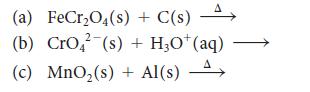
Transcribed Image Text:
(a) FeCr₂O4(s) + C(s) A (b) CrO² (s) + HạO*(aq) (c) MnO₂ (s) + Al(s) A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a Fe 2 O 3 s Cs Fes CO 2 g This is a singledisplacement reaction between ironIII oxide and carbon ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Predict the major products of the following reactions. Include stereochemistry where applicable. (a) 1-methylcycloheptene + BH3 THF, then H2O, OH- (b) Trans-4,4dimethylpent-2ene + BH THF, then H2O2,...
-
Predict the major products of the following reactions: (a) (c) (e) Product of (c) + H2O Product of (c) + H2 (1 molar equivalent) (g) Product of (f) HCI Styrene EtONa HA, heat OH peroxides HA heat Pt...
-
Predict the major products of the following reactions. (a) (E)-3-methyloct-3-ene + ozone, (CH3)2S (b) (Z) -3- methyloct-3ene + warm, concentrated KMnO4 (d) I-ethylcycloheptene +ozone, then (CH3)2S...
-
1 - Describe the purpose of the bubble sort algorithm 2 - The pseudocode below shows the working of the main condition in a bubble sort algorithm, but one part of the code is missinIF items [ i ] >...
-
Look again at the set of efficient portfolios that we calculated in Section 8.1. a. If the interest rate is 10 percent, which of the four efficient portfolios should you hold? b. What is the beta of...
-
The following control procedures are used in Mendy Langs Boutique Shoppe for cash disbursements. 1. The company accountant prepares the bank reconciliation and reports any discrepancies to the owner....
-
Fitted Values. Let \(\left.\widehat{y}_{i}=\pi\left(\mathbf{x}_{i}^{\prime} \mathbf{b}_{M L E} ight) ight)\) denote the \(i\) th fitted value for the logit function. Assume that an intercept is used...
-
Gothic Architecture is a new chain of clothing stores specializing in the color black. Gothic issues 1,000 shares of its $1 par value common stock at $30 per share. Record the issuance of the stock....
-
Identify any five stakeholders who would be interested in Grassland s financial information and explain why and how each would use the information.
-
Name each of the following complex ions and identify the oxidation number of the metal: (a) [CrCl 3 (NH 3 ) 2 (OH 2 )] + ; (b) [Rh(en) 3 ] 3+ ; (c) [Fe(Br) 4 (ox)] 3 ; (d) [Ni(OH)(OH 2 ) 5 ] 2+ .
-
Use the information in Table 9C.1 to write the formula for each of the following coordination compounds: (a) Potassium hexacyanidochromate(III) (b) Pentaamminesulfatocobalt(III) chloride (c)...
-
Evaluate each of the following expressions to six-figure accuracy. (2/5) 3
-
What formula do you use in cell F 2 3 to determine the monthly food budgets in the Budget Table?? For the below budget table, determine the monthly food budgets using the following recuirements...
-
Dunlop Livery Antiques: Modernizing the Vintage Industry Bob Colquhoun, owner of Dunlop Livery Antiques (DLA) in Dunlop, Ontario is developing a comprehensive marketing strategy with a focus on...
-
IE 375 - Production Planning Homework Assignment 1 - October 10 The past demand data for taxi rides at Bilkent Campus is given in the following table. Week 1 Week 2 Week 3 Week 4 Week 5 Week 6 Monday...
-
Question 2 [35 points] CSP- Search Consider a CSP, where there are eight variables A, B, C, D, E, F, G, H, each with domain {1, 2, 3, 4). Suppose the constraints are: A>G |G-C| = 1 D != C G != F...
-
Marigold Industries manufactures three products, Daisy, Rose, and Tulip. Traditionally, the accounting department has allocated indirect costs to four activity pools. Each pool has a unique...
-
Grotto, Inc. exchanged an eight unit apartment building for a four-unit apartment building. Grotto's adjusted basis in the eight-unit building was $320,000 and its fair market value was $400,000. The...
-
Assume a simple Keynesian depression economy with a multiplier of 4 and an initial equilibrium income of $3,000. Saving and investment equal $400, and assume full employment income is $4,000. a. What...
-
Calculate the first five energy levels for a 35 Cl 2 molecule which has a bond length of 198.8 pm if it a. Rotates freely in three dimensions b. Is adsorbed on a surface and forced to rotate in two...
-
An 1 H 35 Cl molecule has the rotational quantum number J = 8 and vibrational quantum number n = 0. a. Calculate the rotational and vibrational energy of the molecule. Compare each of these energies...
-
Calculate the position of the center of mass of a. 1 H 19 F, which has a bond length of 91.68 pm; b. HD, which has a bond length of 74.15 pm.
-
Acctg I Bank Rec Project The Cash account of BCD Associates at June 30, 202X: Beg. Bal Cash 3,995 Check No. 400 Jun 05 102 Jun 06 800 3,010 Jun 12 101 Jun 15 1,800 1,100 Jun 19 103 Jun 23 1,100 500...
-
Choose the best answer that represents the LP formulation for this transportation problem. 5 100 1 6 A 250 200 2 150 50 50 3 3 A 9 6 B 250 4 7
-
The Travel Pro Company sells two kinds of luggage. The company projected the following cost information for the two products: Rolling Carry-on Bag Bag Unit selling price $200 $100 ed Unit variable...

Study smarter with the SolutionInn App


