Use the information in Table 9C.1 to write the formula for each of the following coordination compounds:
Question:
Use the information in Table 9C.1 to write the formula for each of the following coordination compounds:
(a) Potassium hexacyanidochromate(III)
(b) Pentaamminesulfatocobalt(III) chloride
(c) Tetraamminediaquacobalt(III) bromide
(d) Sodium bisoxalato(diaqua)ferrate(III)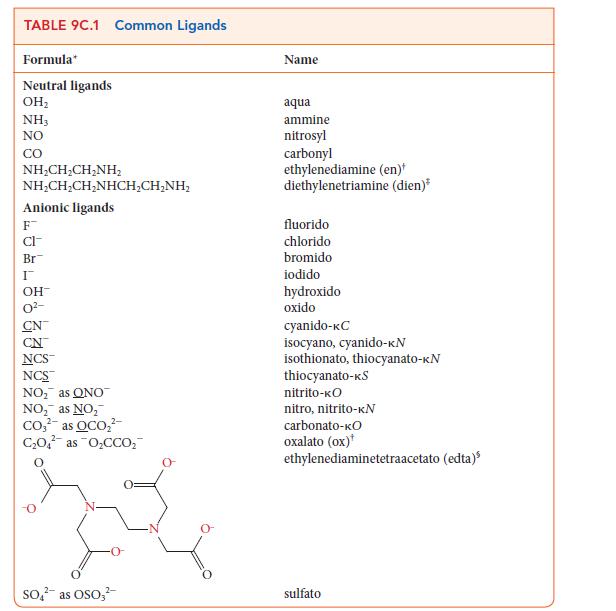
Transcribed Image Text:
TABLE 9C.1 Common Ligands Formula Neutral ligands OH₂ NH3 NO CO NHẠCH,CH_NH, NHẠCH,CH,NHCH,CHÍNH, Anionic ligands F CI- Br I OH- CN NCS™ NCS™ NO₂ as ONO NO₂ as NO₂ Co₂² as OCO₂² C₂O4 as O₂CCO₂ 2- SO² as OSO3² Name aqua ammine nitrosyl carbonyl ethylenediamine (en)* diethylenetriamine (dien)* fluorido chlorido bromido iodido hydroxido oxido cyanido-KC isocyano, cyanido-KN isothionato, thiocyanato-KN thiocyanato-KS nitrito-KO nitro, nitrito-KN carbonato-KO oxalato (ox) ethylenediaminetetraacetato (edta) sulfato
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a KaCrCN6 b ...View the full answer

Answered By

Gabriela Rosalía Castro
I have worked with very different types of students, from little kids to bussines men and women. I have thaught at universities, schools, but mostly in private sessions for specialized purpuses. Sometimes I tutored kids that needed help with their classes at school, some others were high school or college students that needed to prepare for an exam to study abroud. Currently I'm teaching bussiness English for people in bussiness positions that want to improve their skills, and preparing and ex-student to pass a standarized test to study in the UK.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Write the formula for each of the following compounds, being sure to use brackets to indicate the coordination sphere: (a) Hexaamminechromium (III) nitrate (b) Tetraamminecarbonatocobalt (III)...
-
Write the formula for each of the following compounds, being sure to use brackets to indicate the coordination sphere: (a) Tetraaquadibromomanganese(III) perchlorate (b) Bis(bipyridyl)cadmium(II)...
-
Write the chemical formula for each of the following compounds, and indicate the oxidation state of nitrogen in each: (a) Nitric oxide (b) Hydrazine (c) Potassium cyanide (d) Sodium nitrite (e)...
-
Write a program that reads in the x - and y - coordinates in Cartesian space for the endpoints of a line segment and then determines if the line segment is parallel to the y - axis. Hint: a line is...
-
In footnote 4 we noted that the minimum-risk portfolio contained an investment of 21.4 percent in Reebok and 78.6 in Coca-Cola. Prove it.
-
Horvath Company uses an imprest petty cash system. The fund was established on March 1 with a balance of $100. During March, the following petty cash receipts were found in the petty cash box. The...
-
a. Begin with one population and assume that \(y_{1}, \ldots, y_{n}\) is an i.i.d. sample from a Bernoulli distribution with mean \(\pi\). Show that the maximum likelihood estimator of \(\pi\) is...
-
Dorina Company makes cases of canned dog food in batches of 1,000 cases and sells each case for $15. The plant capacity is 50,000 cases; the company currently makes 40,000 cases. DoggieMart has...
-
Audit reporting processses inclusive of "what the report would be against and to whom the report may be accessible to by what methods" and rmore clarification about audit reporting ?
-
Predict the major products of each of the following reactions and then balance the equations: (a) FeCrO4(s) + C(s) A (b) CrO (s) + HO*(aq) (c) MnO (s) + Al(s) A
-
You are analyzing water samples from a local stream and want to use the intense red color of Fe(SCN) 2+ to measure the concentration of Fe 3+ . You need to know the formation constant (Topic 6I) for...
-
How can derivative security be used to hedge portfolio risk?
-
Consider the annual reports of bmw for 2022 uploaded.Using financial ratios, you are required to compare them and answer the following questions:Question 1: For the Balance sheet:* What is the...
-
A single error correcting code, Long(6, 3), is designed so that 3 parity check digits are appended to the end of a 3-digit input, using GF(7). The parity check matrix H is given by 1 1 1 H = 1 2 3 1...
-
Draw a display of the run-time referencing environment, at the point marked "HERE" in the code (5) shown below, for each of the following two techniques: (1) Static Links (2) Displays program main;...
-
III . Daisy and Peter want to purchase a new home and need a loan for $130,000 from the local bank. The loan is for 30 years and the annual interest rate is 4.2%, compounded monthly. They will pay...
-
(1) (2) (3) (4) (5) The extremities of a 8-ft rod weighing 50 lb can move freely and with no friction along two straight tracks as shown. There is an external force (P) applied at G with the...
-
For the Assembly Department, unit materials cost is $8 and unit conversion cost is $12. If there are 8,000 units in ending work in process 75% complete as to conversion costs, the costs to be...
-
The following information is for Montreal Gloves Inc. for the year 2020: Manufacturing costs Number of gloves manufactured Beginning inventory $ 3,016,700 311,000 pairs 0 pairs Sales in 2020 were...
-
Why would you observe a pure rotational spectrum in the microwave region and a rotational vibrational spectrum rather than a pure vibrational spectrum in the infrared region?
-
Solids generally expand as the temperature increases. Such an expansion results from an increase in the bond length between adjacent atoms as the vibrational amplitude increases. Will a harmonic...
-
How can you observe vibrational transitions in Raman spectroscopy using visible light lasers where the photon energy is much larger than the vibrational energy spacing?
-
A company sells a fire pit for $400 per unit and has $45,000 in fixed costs. The variable cost per unit is $325. What is the net income (loss) if the company sells 542 units?
-
Planter Corporation used debentures with a par value of $635,000 to acquire 100 percent of Sorden Company's net assets on January 1, 20X2. On that date, the fair value of the bonds issued by Planter...
-
Estimate the mean of the number of students enrolled in each math class offered at a college given in the following grouped frequency table. Round the final answer to one decimal place. Provide your...

Study smarter with the SolutionInn App


