Rationalize the following boiling points: CH3C OH CICHC CH3C OH OCH3 118C 189C 57C
Question:
Rationalize the following boiling points: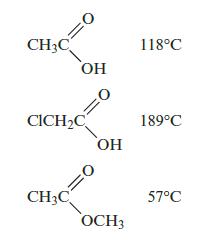
Transcribed Image Text:
CH3C OH CICH₂C CH3C OH OCH3 118°C 189°C 57°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
The boiling points of the compounds can be rationalized as fol...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rationalize the difference in boiling points for each of the following pairs of substances: a. n-pentane CH3CH2CH2CH2CH3 36.2oC b. HF 20oC HCl 85oC c. HCl 85oC LiCl 1360oC d. n-pentane...
-
Rationalize the difference in boiling points in each pair: (a) HF (20 oC) and HCl (- 85 oC), (b) CHCl3 (61 oC) and CHBr3 (150 oC), (c) Br2 (59 oC) and ICI (97 oC)?
-
The boiling points of HCl and CO2 are nearly the same (285C and 278C). Explain why CO2 can be removed from an aqueous solution by boiling briefly while essentially no HCl is lost even after boiling...
-
Water is an essential resource. For that reason moral considerations exert considerable pressure to assure that everyone has access to at least enough water to survive. Yet it appears that equity and...
-
A consumer price analyst claims that prices for liquid crystal display (LCD) computer monitors are normally distributed, with a mean of $190 and a standard deviation of $48. What is the probability...
-
Addition and subtraction. Simplify. x + 0.9x + 0.89x
-
Matching correlations Match each of the following scatterplots to the r below that best describes it. (Some rs will be left over.) r = 0.9 r = 0.7 r = 0.3 r = 0 r = 0.3 r = 0.7 r = 0.9 (b) (d)
-
Knight Corp., a lamp manufacturer, provided the following information for the year ended December 31, 2014. Requirements 1. Use the information to prepare a schedule of cost of goods manufactured. 2....
-
Exercise 10-3 (Algo) Sell or process LO P2 Cobe Company has manufactured 245 partially finished cabinets at a cost of $61,250. These can be sold as is for $73,500. Instead, the cabinets can be...
-
Use the data in the Excel file Baseball Attendance to do the following: a). Develop spreadsheet models for forecasting attendance using single moving average and single exponential smoothing. b)....
-
In February 1997, Chrysler Corporation introduced a new promotional vehicle called the Plymouth Prowler. However, the company did not reveal whether it would manufacture any of the vehicles....
-
What is meant by the term vapor pressure? Why do liquids have a vapor pressure? Do all liquids have a vapor pressure? What about solids? How does vapor pressure change with changing temperature? Why?...
-
Bonnie Goode supervises the online support staff in the customer service department of a software company. The employees are expected to handle 50 phone requests for help per day (250 in a five-day...
-
A new partner C is invited to join in the AB partnership. Currently, A's and B's capital are $540,000 and $100,000, respectively. According to their profit and loss sharing contract, partner A and B...
-
The two tanks shown are connect through a mercury manometer. What is the relation between ???? and ? water Az water Ah
-
1. After reading about the types of rights that prisoners have while incarcerated, which of these rights, if any, should be reduced or diminished? Why? 2. In the same way, what rights do you believe...
-
According to the Socratic view of morality summarized by Frankena, is a person brought up by immoral parents in a corrupt society capable of making correct moral judgements? Why or why not? Do you...
-
Loma Company manufactures basketball backboards. The following information pertains to the company's normal operations per month: Output units15,000 boards Machine-hours4,000 hours Direct...
-
To answer each question, refer to the following basic graphs. Which one is the graph of (x) = 0 x 0 ? On what open interval is it increasing? C. A. B. 123 -8 F. E. D. Ho G. . I. 8. 2- -2 8. -2 2.
-
Why did management adopt the new plan even though it provides a smaller expected number of exposures than the original plan recommended by the original linear programming model?
-
Using bond energies from Table, estimate the barrier to rotation around a carboncarbon double bond. To do this, consider what must happen to go from In terms of making and breaking chemical bonds;...
-
Show how a dxz atomic orbital and a pz atomic orbital combine to form a bonding molecular orbital. Assume the x axis is the internuclear axis. Is a or a molecular orbital formed? Explain.
-
Describe the bonding in the first excited state of N2 (the one closest in energy to the ground state) using the MO model. What differences do you expect in the properties of the molecule in the...
-
QUESTION 3 A business owns seven flats rented out to staff at R500 per month. All flats were tenanted Ist january 21 months rent was in arrears and as at 31st December 14 months' rent wa Identify the...
-
1. 2. 3. Select the Tables sheet, select cells A6:B10, and create range names using the Create from Selection button [Formulas tab, Defined Names group]. Select cells B1:F2 and click the Name box....
-
Tropical Rainwear issues 3,000 shares of its $18 par value preferred stock for cash at $20 per share. Record the issuance of the preferred shares. (If no entry is required for a particular...

Study smarter with the SolutionInn App


