Some entries for S m in Appendix 2A are negative. What is common about these entries,
Question:
Some entries for Sm° in Appendix 2A are negative. What is common about these entries, and why would the entropy be negative?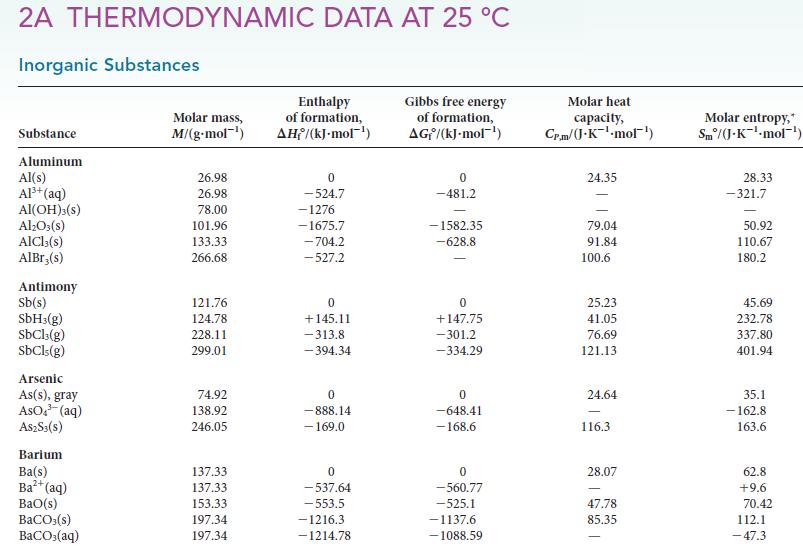
Transcribed Image Text:
2A THERMODYNAMIC DATA AT 25 °C Inorganic Substances Substance Aluminum Al(s) Al³+ (aq) Al(OH)3(S) Al₂O3(s) AlCl3(s) AlBr,(s) Antimony Sb(s) SbH3(g) SbCl3(g) SbCls (g) Arsenic As(s), gray AsO³(aq) A$2S3(S) Barium Ba(s) Ba²+ (aq) BaO(s) BaCO3(s) BaCO3(aq) Molar mass, M/(g.mol-¹) 26.98 26.98 78.00 101.96 133.33 266.68 121.76 124.78 228.11 299.01 74.92 138.92 246.05 137.33 137.33 153.33 197.34 197.34 Enthalpy of formation, AH/(kJ-mol-¹) 0 -524.7 -1276 -1675.7 -704.2 -527.2 0 +145.11 -313.8 -394.34 0 -888.14 - 169.0 0 -537.64 -553.5 -1216.3 -1214.78 Gibbs free energy of formation, AG/(kJ.mol-¹) 0 -481.2 -1582.35 -628.8 0 +147.75 -301.2 -334.29 0 -648.41 -168.6 0 -560.77 -525.1 -1137.6 -1088.59 Molar heat capacity, Cr.m/J.K¹-mol¹) 24.35 79.04 91.84 100.6 25.23 41.05 76.69 121.13 24.64 116.3 28.07 47.78 85.35 Molar entropy, Sm/(J-K¹-mol-¹) 28.33 -321.7 50.92 110.67 180.2 45.69 232.78 337.80 401.94 35.1 -162.8 163.6 62.8 +9.6 70.42 112.1 -47.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The entries all correspond to aqueous ions The fact that they are negative is due to the reference ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
The Professional Judgment in Context scenario at the beginning of the chapter introduced excerpts from the PCAOB inspection of Deloitte & Touche, LLP. The scenario is: On May 4, 2010, the PCAOB...
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
On May 4, 2010, the PCAOB issued its public inspection of Deloitte & Touche, LLP, covering their inspection of audits conducted during 2009. In their summary comments, the PCAOB inspectors stated: In...
-
The chief accountant for Dickinson Corporation provides you with the following list of accounts receivable that were written off in the current year: Dickinson Corporation follows the policy of...
-
A two-dimensional potential exists on a unit square area (0 ? x ? l, 0 ? y ? l) bounded by "surfaces" held at zero potential. Over the entire square there is a uniform charge density of unit strength...
-
Draw a sketch of the region in which the points satisfy the given inequality or system of inequalities. y > 12 3x
-
For each of the following processes, determine what energy conversion takes place and classify the interaction as dissipative or nondissipative. (a) The launching of a ball by the expanding of a...
-
Cosmotologist Warehouse distributes commercial hair care products in one-gallon bottles to hair salons and extends credit terms of 3/10, n/30 to all of its customers. During the month of April, the...
-
15 16 17 18 In 1987, an agreement was formulated by the United Nations Environment Programme (UNEP) to freeze the production of "X" to prevent depletion of "Y". "X" and "Y" respectively referred here...
-
Three isomeric alkenes have the formula C 4 H 8 (see the following table). (a) Draw Lewis structures of these compounds. (b) Calculate G, H, and S for the three reactions that interconvert each pair...
-
Acetic acid, CH 3 COOH(l), could be produced from (a) The reaction of methanol with carbon monoxide; (b) The oxidation of ethanol; (c) The reaction of carbon dioxide with methane. Write balanced...
-
Another useful exercise is to ask individuals to rate their own performance. Using the performance appraisal instrument at your current employer (or former place of employment, or the instrument...
-
Suppose your wealthy aunt gave you and your cousins \(\$ 10000\) each. Assume for a moment that you are not associated with Sweet Temptations and that you are considering loaning the \(\$ 10000\) to...
-
The Huron Company keeps a petty cash fund of \(\$ 80\). On 30 June the fund contained cash of \(\$ 36.87\) and these petty cash receipts: Required: a If the business' fiscal year ends 30 June should...
-
On 1 January, the balance in the Accounts Receivable Account for James Pty Ltd was \(\$ 4125\). During the next six months, credit sales amounted to \(\$ 13075\) and clients paid \(\$ 14560\) to...
-
On 31 December the Big Hole Property Management Company had a balance of \(\$ 70\) in its petty cash fund, a reconciled balance of \(\$ 1283\) in its cheque account and a \(\$ 4627\) balance in its...
-
The Gomez Folding Chair Company has 400 chairs (at \(\$ 15\) each) in its beginning inventory for July. It makes the following purchases and sales of chairs during July: Required: Calculate the cost...
-
Put yourself in the yacht dealers shoes. You currently are considering other order quantities in addition to 50 and 100. Find the optimal order quantity, that is, the exact quantity that maximizes...
-
What are the 5 Cs of marketing channel structure?
-
Which of the following is the best reducing agent: F 2 , H + , Na, Na + , or F 2 ? Explain. Order as many of these species as possible from the best to the worst oxidizing agent. Why cant you order...
-
Which of the following is the best reducing agent: F 2 , H + , Na, Na + , or F 2 ? Explain. Order as many of these species as possible from the best to the worst oxidizing agent. Why cant you order...
-
Sketch a galvanic cell, and explain how it works. Look at Figs. 11.1 and 11.2. Explain what is occurring in each container and why the cell in Fig. 11.2 works but the one in Fig. 11.1 does not. Fig....
-
The position of a particle traveling along a horizontal line in t seconds is s(t) = 2+3 - 12t - 126+ + 6 meters. (a). Find the velocity and acceleration functions at time t = 3 seconds. (b). Is the...
-
A 200 g copper wire loses 1.6 KJ of thermal energy to its surroundings. What is the change in temperature?
-
= 17: Doppler shift of a spectral line: One of the most prominent spectral lines of hydrogen is the H line, a bright red line with a wavelength of \o 656.1 10-9 m. Note: The speed c, wavelength \ and...

Study smarter with the SolutionInn App


