Some of the physical properties of (mathrm{H}_{2} mathrm{O}) and (mathrm{D}_{2} mathrm{O}) are as follows: Account for the
Question:
Some of the physical properties of \(\mathrm{H}_{2} \mathrm{O}\) and \(\mathrm{D}_{2} \mathrm{O}\) are as follows:
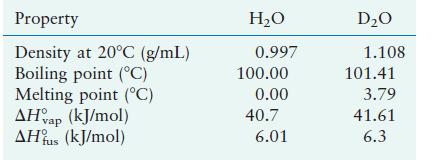
Account for the differences. (D is a symbol often used for \({ }^{2} \mathrm{H}\), the deuterium isotope of hydrogen.)
Transcribed Image Text:
Property Density at 20C (g/mL) Boiling point (C) Melting point (C) AHvap (kJ/mol) AHus (kJ/mol) HO 0.997 100.00 0.00 40.7 6.01 DO 1.108 101.41 3.79 41.61 6.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The differences in the physical properties between H2O water and D2O heavy water can be accounted fo...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The formula for shallow-water wave propagation speed, Eq. (10.9) or (10.10), is independent of the physical properties of the liquid, i.e., density, viscosity, or surface tension. Does this mean that...
-
When training a group of workers, often the challenge for the trainer is ensuring the key messages are getting through to everyone. In larger groups, its not always easy keeping everyone engaged, and...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family. The Incisors own a rental beach house in Hawaii. The beach house was rented for the full year during 2012...
-
Consider the following molecule: Tin II Chloride(SnCl2) Are the bonds polar or non-polar? Is the molecule polar or non-polar?
-
A tank contains 1000 L of pure water. Brine that contains 0.05 kg of salt per liter of water enters the tank at a rate of 5 L/min. Brine that contains 0.04 kg of salt per liter of water enters the...
-
Fluorescent lights contain spectral lines from the light emission of mercury atoms. Special coatings on the inner surface of the bulb help to accentuate visible frequencies, which can be seen through...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. Determine the ERR for this project. b. Is this project economically attractive? EOY 0 2 3 4 5 6 NCF -$100 $15 $15 $15...
-
During Heaton Companys first two years of operations, the company reported absorption costing net operating income as follows: The companys $18 unit product cost is computed as follows: Forty percent...
-
1) Write a Python function to convert mass values from gram to ounce and return the conversion result. The conversion formula is ounce = gram x 0.035274. The function header is: def gm_to_ounce...
-
The unit cell for a pure xenon fluoride compound is shown in the following diagram. a. What is the formula of the compound? b. The unit cell of \(\mathrm{XeF}_{2}\) has a height of \(702...
-
Consider the following compounds and formulas. (The formulas are written in such a way as to give you an idea of the structure.) The boiling points of these compounds are (in no particular order)...
-
A particle with charge 2.0 C moves through a uniform magnetic field. At one instant the velocity of the particle is (2.0 i + 4.0 j + 6.0 k) m/s and the magnetic force on the particle is (4.0 i 20 j...
-
Josh Lee has owned a fitness center for the past four years. He has historical financial statements but has never put together a set of pro forma financial statements. He just applied for a bank loan...
-
Write a mission statement for 99designs, the company highlighted in the Partnering for Success feature. How can this statement help clarify and direct the core strategy component of 99designs...
-
Assume that Shoaib Shaikh, the founder of XploSafe (the company focused on at the beginning of the chapter), has asked you to help him write a code of conduct for his firm. Given your understanding...
-
What are the two primary functions of the financial management of a firm?
-
Describe the purpose of having an organized sales process.
-
The company issued convertible bonds with a total face value of $100,000 for $107,000. If the bonds had been issued without the conversion feature, their issuance price would have been $98,000. Make...
-
Determine the optimal use of Applichem's plant capacity using the Solver in Excel.
-
When iron surfaces are exposed to ammonia at high temperatures, nitridingthe incorporation of nitrogen into the iron latticeoccurs. The atomic radius of iron is 124 pm. (a) Is the alloy interstitial...
-
How would the height of a mercury barometer be different on the planet Mars? Explain your reasoning.
-
Air is a source of reactants for many chemical processes, such as the synthesis of ammonia. To determine how much air is needed for these reactions, it is useful to know the partial pressures of the...
-
Janice bought her house in 2011 for $395,000. Since then, she has deducted $70,000 in depreciation associated with her home office and has spent $45,000 replacing all the old pipes and plumbing. She...
-
In October, Blossom Company reports 18,100 actual direct labor hours, and it incurs $100,500 of manufacturing overhead costs. Standard hours allowed for the work done is 20,100 hours. The...
-
Leslie Sporting Goods is a locally owned store that specializes in printing team jerseys. The majority of its business comes from orders for various local teams and organizations. While Leslie's...

Study smarter with the SolutionInn App


