The equilibrium constant for the following reaction is (1.0 times 10^{23}) : EDTA is used as a
Question:
The equilibrium constant for the following reaction is \(1.0 \times 10^{23}\) :
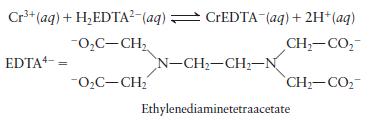
EDTA is used as a complexing agent in chemical analysis. Solutions of EDTA, usually containing the disodium salt \(\mathrm{Na}_{2} \mathrm{H}_{2}\) EDTA, are used to treat heavy metal poisoning. Calculate \(\left[\mathrm{Cr}^{3+}ight]\) at equilibrium in a solution originally \(0.0010 \mathrm{Min}^{3+}\) and \(0.050 \mathrm{M}\) in \(\mathrm{H}_{2}\) EDTA \(^{2-}\) and buffered at \(\mathrm{pH}=6.00\).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





