The table below lists the ionic radii for the cations and anions in three different ionic compounds.
Question:
The table below lists the ionic radii for the cations and anions in three different ionic compounds.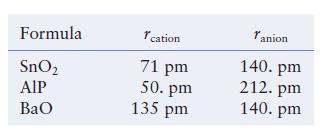 Each compound has either the NaCl, CsCl, or ZnS type cubic structure. Predict the type of structure formed (NaCl, CsCl, or ZnS) and the type and fraction of holes filled by the cations, and estimate the density of each compound.
Each compound has either the NaCl, CsCl, or ZnS type cubic structure. Predict the type of structure formed (NaCl, CsCl, or ZnS) and the type and fraction of holes filled by the cations, and estimate the density of each compound.
Transcribed Image Text:
Formula SnO₂ AIP BaO cation 71 pm 50. pm 135 pm Tanion 140. pm 212. pm 140. pm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 28% (7 reviews)
the type of structure formed is ZnS Each compound has either the NaCl CsCl or ZnS type cubic structu...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The table below lists the ionic radii for the cations and anions in three different ionic compounds. Each compound has either the NaCl, CsCl, or ZnS type cubic structure. Predict the type of...
-
The rutile and fluorite structures, shown here (anions are colored green), are two of the most common structure types for ionic compounds where the cation to anion ratio is 1:2. (a) For CaF2 and ZnF2...
-
NaCl and KF have the same crystal structure. The only difference between the two is the distance that separates cations and anions. (a) The lattice energies of NaCl and KF are given in Table 8.2....
-
Suppose there are two identical forest plots except that one will be harvested and left as is while the second will be cleared after the harvest and turned into a housing development. In terms of...
-
The annual per capita consumption of canned vegetables by people in the United States is normally distributed, with a mean of 39 pounds and a standard deviation of 3.2 pounds. Random samples of size...
-
A disk (Idisk = 12MR2) of mass 200kg and radius 2m is rotating at rad/s. A small object of mass 40 kg then collides with and sticks to the disk at a location halfway between the center and the edge....
-
The force transmitted by an internal combustion engine of mass \(500 \mathrm{~kg}\), when placed directly on a rigid floor, is given by \[F_{t}(t)=(18,000 \cos 300 t+3600 \cos 600 t) \mathrm{N}\]...
-
The trial balance of Marius Santiago Co. shown below does not balance. Each of the listed accounts has a normal balance per the general ledger. An examination of the ledger and journal reveals the...
-
Hardeep Shah has recently been appointed as the new Company Secretary of Teroja Finance Berhad, a company that provides financial advisory services, which has its shares listed on the Bursa Malaysia....
-
The phrase scientia est potentia is a Latin aphorism meaning knowledge is power! In a business sense, this might be paraphrased as Information is money! Better information . . . better business...
-
How does DHsoln relate to deviations from Raoults law? Explain.
-
What type of solid (network, metallic, Group 8A, ionic, or molecular) will each of the following substances form? a. Kr b. SO 2 c. Ni d. SiO 2 e. NH 3 f. Pt
-
Define the following terms: genomics, functional genomics, and proteomics.
-
List and briefly describe the golden rules for success in formal negotiations.
-
What are the reasons for a follow-up with customers, and why do they add value?
-
Why is good cost allocation critical to good decision-making?
-
A country with a persistent trade surplus is being pressured to let its currency appreciate. Which of the following best describes the adjustment that must occur if currency appreciation is to be...
-
What are the two most important points to keep in mind when dealing with price resistance?
-
On rare occasions, people are born with a condition known as uniparental disomy. It happens when an individual inherits both copies of a chromosome from one parent and no copies from the other...
-
Calculate the change in entropy when 100 kJ of energy is transferred reversibly and isothermally as heat to a large block of copper at (i) 0 C, (ii) 50 C.
-
Give the expected hybridization of the central atom for the molecules or ions in Exercises 57, 58, and 60 from Chapter 13.
-
Give the expected hybridization of the central atom for the molecules in Exercises 91 and 92 from Chapter 13.
-
Urea, a compound formed in the liver, is one of the ways humans excrete nitrogen. The Lewis structure for urea is Using hybrid orbitals for carbon, nitrogen, and oxygen, determine which orbitals...
-
We have initially touched on some critical elements of an IT security program through our readings, class discussions, and class lectures. For this discussion provide a substantive post depicting...
-
On January 2, Year 4, Brady Ltd., a private company, purchased 80% of the outstanding shares of Partridge Ltd. for $5,420,000. Partridge's statement of financial position and the fair values of its...
-
Read string fav_color from input, and output the following, all separated by spaces: 'My favorite color' The value of fav_color 'contains' The length of fav_color 'characters ?

Study smarter with the SolutionInn App


