The two strands of the nucleic acid DNA are held together by hydrogen bonding between four organic
Question:
The two strands of the nucleic acid DNA are held together by hydrogen bonding between four organic bases. The structure of one of these bases, thymine, is shown below.
(a) How many protons can this base accept?
(b) Draw the structure of each conjugate acid that can be formed.
(c) Mark with an asterisk any structure that can show amphiprotic behavior in aqueous solution.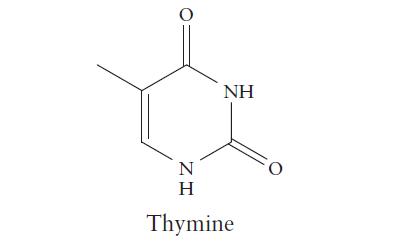
Transcribed Image Text:
O N ZH Η NH Thymine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a Two protons can be accepted b O o...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
The two strands of the nucleic acid DNA are held together by hydrogen bonding between four organic bases. The structure of one of these bases, cytosine, is shown below. (a) How many protons can this...
-
The two strands of the helix-shaped DNA molecule are held together by electrostatic forces as shown in Fig. 16-44. Assume that the net average charge (due to electron sharing) indicated on H and N...
-
Figure 1. Four different bonds (W, X, Y, and Z) in a DNA molecule Figure 1 represents a segment of DNA. Radiation can damage the nucleotides in a DNA molecule. To repair some types of damage, a...
-
The following sequences of bits (rightmost bit first) appear at the inputs of a 4-bit parallel adder. Determine the resulting sequence of bits on each output. A1 1 0 0 1 A2 1 1 1 0 A3 0 0 0 0 A4 1 0...
-
Theory of constraints, throughput contribution, relevant costs. The Mayfield Corporation manufactures filing cabinets in two operations: machining and finishing. It provides the following...
-
Listed below are several terms and phrases associated with current liabilities. Pair each item from List A (by letter) with the item from List B that is most appropriately associated withit. List A...
-
Under IFRS, for non-financial companies, most non-current assets are measured at: A. Fair value. B. The lower of cost or fair value. C. Cost less depreciation and impairment. D. Net realisable value.
-
George Company purchased land for use as its corporate headquarters. A small factory that was on the land when it was purchased was torn down, and before the new buildings foundation could be...
-
1. Let be the vector : err =[0.5671; 0.4328%; 0.4555e-01; 0.3305e-02; 0.2707e-04; 0.1660e-7]; In PYTHON, we want to calculate the values of the ratios : errn+1 errn errn+1 errn errn+1 et err 1+5...
-
Which of the following indicators in Table 6H.2 could you use for a titration of 0.20 m CH 3 COOH(aq) with 0.20 m NaOH(aq): (a) Methyl orange; (b) Litmus; (c) Thymol blue; (d) Phenolphthalein?...
-
Is the osmotic pressure of 0.10 m H 2 SO 4 (aq) the same as, less than, or greater than that of 0.10 m HCl(aq)? Calculate the osmotic pressure of each solution to support your conclusion.
-
She marketing manager used shelf space for pet food to predict weekly sales. The data are stored in Pet food. From the results of that problem, b1 = 7.4 and Sbl = 1.59. a. At the 0.05 level of...
-
When cash is invested to earn a higher return than a bank savings account, what are the two primary tradeoffs?
-
Suppose that you need 5,000 units of inventory over the course of a year. Each unit costs $\$ 1$. The costs related to placing each order are $\$ 20$. Your cost of capital is $8 %$. Other carrying...
-
What are the three principal reasons that organizations keep cash?
-
Account payable are sometimes referred to as .
-
What are the variables that have to be considered in the EOQ system for inventory?
-
Many people believe that the budgeting process is wasteful and ineffective. They maintain that managers spend too much time focusing on the mechanics of budgeting and not enough time on strategic...
-
By referring to Figure 13.18, determine the mass of each of the following salts required to form a saturated solution in 250 g of water at 30 oC: (a) KClO3, (b) Pb(NO3)2, (c) Ce2(SO4)3.
-
When 2-ethyl-5-chlorotoluene was treated with sodium hydroxide at high temperature, followed by treatment with H 3 O + , three constitutional isomers with molecular formula C 9 H 12 O were obtained....
-
You wish to design an effusion source for Br atoms from Br 2 (g). If the source is to operate at a total pressure of 7.5 Torr, what temperature is required to produce a degree of dissociation of...
-
Calculate G for the isothermal expansion of 2.25 mol of an ideal gas at 325 K from an initial pressure of 12.0 bar to a final pressure of 2.5 bar.
-
AndrIt iss dissolved 36.167gSn(C2H302), in enough distilled water to make 200.0mL of solution. Perform the following calculations regarding this solution: A. What is the molarity of this solution?...
-
If a Person On a specific diet must include all three foods in their daily intake Of 2480 Calories, 114 grams of protein, and 175 M of Sodium, how many ounces of each food can the person eat for day?...
-
Using the appropriate bond energies, calculate the heat of reaction AH for the following reaction: H | 2H C H + 30=0 H 2C=0 + 40-H H You can find a table of bond energies by using the Data button on...

Study smarter with the SolutionInn App


