Which of the following indicators in Table 6H.2 could you use for a titration of 0.20 m
Question:
Which of the following indicators in Table 6H.2 could you use for a titration of 0.20 m CH3COOH(aq) with 0.20 m NaOH(aq):
(a) Methyl orange;
(b) Litmus;
(c) Thymol blue;
(d) Phenolphthalein?
Explain your selections.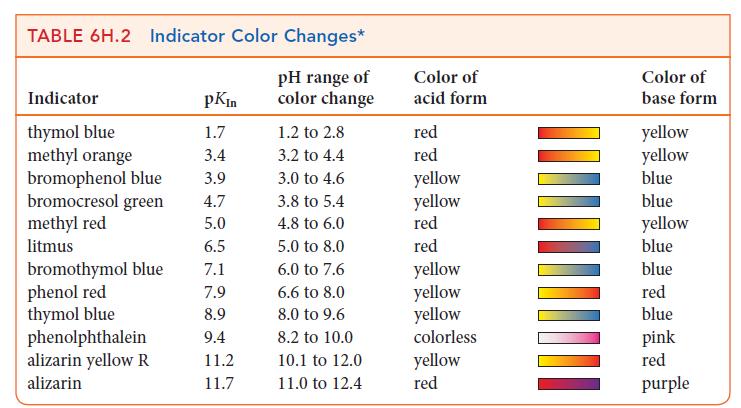
Transcribed Image Text:
TABLE 6H.2 Indicator Color Changes* pH range of color change Indicator thymol blue methyl orange bromophenol blue bromocresol green methyl red litmus bromothymol blue phenol red thymol blue phenolphthalein alizarin yellow R alizarin pKin 1.7 3.4 3.9 4.7 5.0 6.5 7.1 7.9 8.9 9.4 11.2 11.7 1.2 to 2.8 3.2 to 4.4 3.0 to 4.6 3.8 to 5.4 4.8 to 6.0 5.0 to 8.0 6.0 to 7.6 6.6 to 8.0 8.0 to 9.6 8.2 to 10.0 10.1 to 12.0 11.0 to 12.4 Color of acid form red red yellow yellow red red yellow yellow yellow colorless yellow red Color of base form yellow yellow blue blue yellow blue blue red blue pink red purple
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
d Phenolph...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Which of the following indicators in Table 6H.2 could you use for a titration of 0.20 m NH 3 (aq) with 0.20 m HCl(aq): (a) Bromocresol green; (b) Methyl red; (c) Phenol red; (d) Thymol blue? Explain...
-
You would like to invest in one of the three available investment plans: money market, bonds, or stocks. The payoffs (profits) of each plan under two possible future economic conditions, PE (poor...
-
Procedures Experiment 1: Standardize the Iodine Solution Part 1: Prepare the Materials Take a 100 mL volumetric flask from the Containers shelf and place it on the workbench. Take ascorbic acid from...
-
Sunland Company has the below information for accruals for the year ended December 31st, 2022. the company adjusts its accounts annually. Chapter 4 Homework e Question 6 of 7 Current Attempt in...
-
Theory of constraints, throughput contribution, quality. Refer to the information in Exercise 19-25 in answering the following requirements. There is no connection between the requirements. 1....
-
Lee Financial Services pays employees monthly. Payroll information is listed below for January 2011, the first month of Lees fiscal year. Assume that none of the employees exceeded any relevant wage...
-
Provide the ideal gain scheduling for Figure 12.16 as a function of Q. DATA FROM FIGURE 12.16 Assume inertia varies with theta as follows: where JM is the motor inertia, JL is the load inertia, and R...
-
Johan recently received his annual performance bonus from his employer. He has set up an investment savings plan to which he will contribute $2000 each year from his bonus and $400 per month from his...
-
2 If logx2 = 1.2a and logx3 = 1.5a, in terms of a, find the answer to logx 12
-
The two strands of the nucleic acid DNA are held together by hydrogen bonding between four organic bases. The structure of one of these bases, cytosine, is shown below. (a) How many protons can this...
-
The two strands of the nucleic acid DNA are held together by hydrogen bonding between four organic bases. The structure of one of these bases, thymine, is shown below. (a) How many protons can this...
-
Answer true or false to each statement, and explain your answers. a. The y-intercept of a line has no effect on the steepness of the line. b. A horizontal line has no slope. c. If a line has a...
-
Compute the following quantity using MATLAB in the Command Window: \[B=\frac{\tan x+\sin 2 x}{\cos x}+\log \left|x^{5}-x^{2} ight|+\cosh x-2 \tanh x\] for $x=5 \pi / 6$.
-
Assume that a 30 year, $5 %$, semiannual, $\$ 10,000$ bond was issued ten years ago. Today current market rates are $4.5 %$. How much could that bond be sold for today?
-
What is net working capital, or simply working capital?
-
What are the steps from issuance of a bill until we collect payment from a customer and have access to the funds? What role do managers play, aside from issuing the bill?
-
What is the period of time from which you write a check until it clears your bank account called?
-
Suppose you have just signed a partnership agreement with your cousin Eddie to open a bookstore near your college. You believe that the store will be able to provide excellent service and undersell...
-
The nitrogen atoms in N2 participate in multiple bonding, whereas those in hydrazine, N2H4, do not. (a) Draw Lewis structures for both molecules. (b) What is the hybridization of the nitrogen atoms...
-
Predict the change in the partial pressure of CO 2 as the pressure is increased at constant temperature. CO(g) + 1/2O 2 (g) CO 2 (g) at equilibrium for which H o R = 283.0 kJ mol 1 .
-
Predict the change in the partial pressure of CO 2 as the pressure is increased at constant temperature. CO(g) + 1/2O 2 (g) CO 2 (g) at equilibrium for which H o R = 283.0 kJ mol 1 .
-
Predict the change in the partial pressure of CO 2 as Xe gas is introduced into the reaction vessel at constant pressure and temperature. CO(g) + 1/2O 2 (g) CO 2 (g) at equilibrium for which H o R =...
-
By definition, an important aspect of culture is that it is living and living things evolve and change over time; they leave legacies. For this week, please discuss the connections you see between...
-
Consider the following polynomial functions. g(x)=-2x+8x h(x)=(x+4) (x-1)
-
3. For various combination quantum numbers I, mi and ms, enter the appropriate information in the following table. 4. mi I ms Type of orbital Total number of orbitals of this type Draw an outline of...

Study smarter with the SolutionInn App


