Use data from Appendix 2B to decide whether chlorine gas will oxidize Mn 2+ to form the
Question:
Use data from Appendix 2B to decide whether chlorine gas will oxidize Mn2+ to form the permanganate ion in an acidic solution.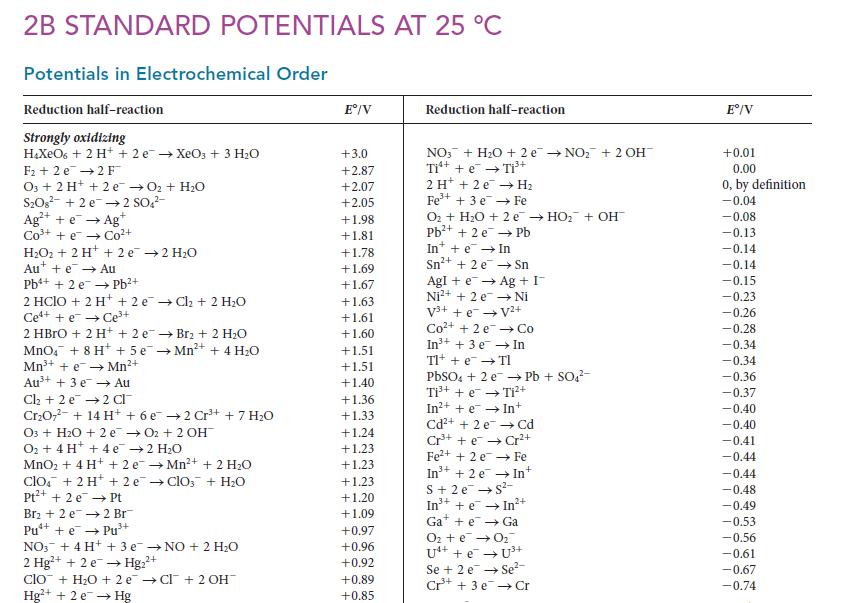
Transcribed Image Text:
2B STANDARD POTENTIALS AT 25 C Potentials in Electrochemical Order Reduction half-reaction Strongly oxidizing HXeO6 + 2 H+ + 2 e XeO3 + 3 HO F+2 e 2 F O3 + 2 H+ + 2e O + HO SO8 +2e 2 SO4- Ag+ + e Ag C t HO + 2H+ + 2e 2 HO Aue Au Pb+ + 2 e Pb+ 2 HClO + 2 H+ + 2 eCl + 2 HO Ce++eCe+ 2 HBrO + 2H+ + 2 e Br2 + 2 HO MnO4 + 8 H+ + 5e Mn+ + 4HO Mn+ + eMn+ Au+ + 3 e Au Cl + 2 e 2 Cl CrO7- + 14 H+ + 6 e cot O3 + HO + 2 e O + 4H+ + 4e 2 HO MnO + 4H+ + 2e Mn+ + 2 HO clo + 2 H+ + 2 eClO3 + HO Pt+ + 2e Pt Br + 2 e Pu+ + ePu+ 2 Br 3+ 2 Cr+ + 7 HO O2 + 2 OH NO3 + 4H+ + 3 eNO + 2 HO 2 Hg+ + 2e Hg+ 2+ clo + HO +2e Cl + 2 OH Hg+ + 2e Hg E/V +3.0 +2.87 +2.07 +2.05 +1.98 +1.81 +1.78 +1.69 +1.67 +1.63 +1.61 +1.60 +1.51 +1.51 +1.40 +1.36 +1.33 +1.24 +1.23 +1.23 +1.23 +1.20 +1.09 +0.97 +0.96 +0.92 +0.89 +0.85 Reduction half-reaction NO3 + HO + 2e NO + 2 OH- Ti+ + e Ti+ 2 H + 2e H Fe+ + 3 e Fe O + HO + 2e HO + OH Pb+ + 2 e Pb In+ + e In Sn+ + 2 e Sn Ag + I Ni AgI + e Ni+ + 2 e V+ + e V+ Co+ + 2e In+ + 3 e Tl+ + e TI Co In PbSO4 + 2 ePb + SO4- Ti+ + e Ti+ In++eIn+ Cd+ + 2 e Cd Cr+ + eCr+ Fe+ + 2e Fe In +2 e S+ 2e S- + In +eIn2+ Gae Ga O + e 0 U4+ + e Se + 2 eSe- Cr+ + 3e Cr E/V +0.01 0.00 0, by definition -0.04 -0.08 -0.13 -0.14 -0.14 -0.15 -0.23 -0.26 -0.28 -0.34 -0.34 -0.36 -0.37 -0.40 -0.40 -0.41 -0.44 -0.44 -0.48 -0.49 -0.53 -0.56 -0.61 -0.67 -0.74
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
2 Clg will n...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Use electrode potentials to answer the following questions, assuming standard conditions. (a) Do you expect permanganate ion (MnO4 ) to oxidize chloride ion to chlorine gas in acidic solution? (b)...
-
A customer made a reservation via telephone for a two-double bed room at the hotel. Unfortunately, once we arrived at the hotel, there was an error with the reservation and there were no rooms left...
-
(a) Use the data in Appendix 2B to decide which of ozone and fluorine is the stronger oxidizing agent in water. (b) Does youranswer depend on whether the reaction is carried out in acidic or basic...
-
The net income for Rips Curling Corp. for the year ended December 31, 2021, was $700,000. Rip had 50,000 ordinary shares outstanding at the beginning of the year. Rip declared and distributed a...
-
After genetically engineering a bacterium capable of breaking down crude oil, Ananda Chakrabarty sought to patent his creation under Title 35 U.S. Code Section 101, which states that "[w]hoever...
-
Let \(\mathbf{F}\) be a vector field whose curl and divergence at the origin are \[ \operatorname{curl}(\mathbf{F})(0,0,0)=\langle 2,-1,4angle, \quad \operatorname{div}(\mathbf{F})(0,0,0)=-2 \]...
-
Sabrina Ederly worked as a reporter and contributing editor for Rolling Stone magazine. Ederly intended to write an article for the magazine that focused on the alleged rape of a University of...
-
The Tall Oaks Wood Products Company is considering purchasing timberland for $5 million that would provide a future source of timber supply for the companys operations over the next 10 years....
-
Three children must make fair division of a painting and sculpture left them by their mother. Using the Knaster Inheritance Procedure, the value each child places on the objects is shown below....
-
Isoelectronic species have the same number of electrons. (a) Divide the following species into three isoelectronic groups: NH 3 , NO, NO 2 + , N 2 O, H 3 O + , O2 + . (b) Which species in each group...
-
Identify the products and write a balanced equation for the reaction of hydrogen with (a) Chlorine; (b) Sodium; (c) Phosphorus; (d) Copper.
-
Calculate the mass percentage of copper in CuS, copper(II) sulfide.
-
Part I: Answer the following questions 1. There are several strategies to persuade your audience. Discuss one strategy in detail and provide an example of how you could use this strategy to persuade...
-
A . Hearts had a net income of $ 4 7 5 , 0 0 0 . The company only uses cash dividends. B . Hearts bought $ 7 4 , 0 0 0 worth of investments and sold other investments for a gain of $ 8 , 3 0 0 . C ....
-
Convert the coordinate vector [x] from the given basis B to the standard basis. B = {[] []}, = [2] x=
-
As a Sr. Technical Writer, you have been tasked to train a team of communicators about how to utilize visual and textual elements in documentation effectively. Please include all the information in...
-
Implement the following two recursive algorithms. The power function, p(x,n)=x", can be defined recursively as: 1. 2. p(x,n)= ;n)= = {x-p(x, 1 if n = 0 (x.p(x,n1) else 1 if x = 0 p(x,n)=x...
-
The nuclide 198Au, with a half-life of 2.70 d, is used in cancer therapy. What mass of this nuclide is required to produce an activity of 250 Ci?
-
The comparative statements of financial position of Menachem NV at the beginning and end of the year 2019 appear below. Net income of ¬34,000 was reported, and dividends of ¬23,000 were paid...
-
The shear stressstrain diagram for a solid 50-mm-diameter shaft can be approximated as shown in the figure. Determine the torque required to cause a maximum shear stress in the shaft of 125 MPa. If...
-
A steel alloy core is bonded firmly to the copper alloy tube to form the shaft shown. If the materials have the Ï-γ diagrams shown, determine the torque resisted by the core and the...
-
The shaft consists of two sections that are rigidly connected. If the material is elastic perfectly plastic as shown, determine the largest torque T that can be applied to the shaft. Also, draw the...
-
solve Given g(x, y, z) = ez sin(4x) cos(y), Vg =
-
2. Your For a Bangor, Maine area route design project, you have performed astronomic observations for directional control at a series of places. boss tells you since it is a small area the same...
-
dg Find dg dt dt where g(x, y) = xy and x = sin(t), y = ln(t). Reduce your answer to one variable.

Study smarter with the SolutionInn App


