Use data from Tables 6C.1 and 6C.2 to place the following acids in order of increasing strength:
Question:
Use data from Tables 6C.1 and 6C.2 to place the following acids in order of increasing strength: HNO2, HClO2, +NH3OH, (CH3)2NH2+.
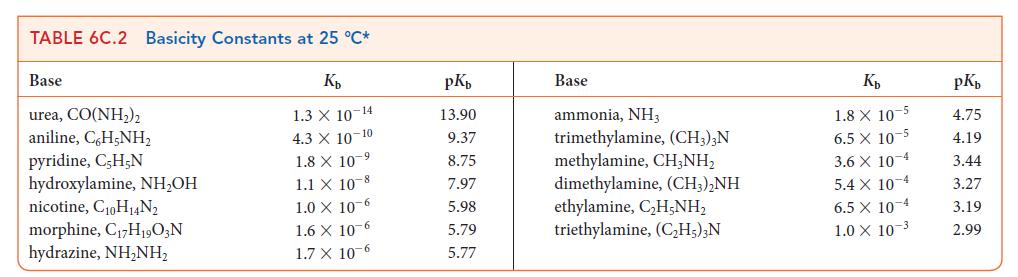
Transcribed Image Text:
TABLE 6C.1 Acidity Constants at 25 °C* K₂ 3.0 X 10-1 2.0 X 10-1 1.7 X 10-1 1.5 X 1.0 X 10-2 Acid trichloroacetic acid, CCI,COOH benzene sulfonic acid, C,H,SO3H iodic acid, HIO3 sulfurous acid, H₂SO3 chlorous acid, HClO₂ phosphoric acid, H3PO4 chloroacetic acid, CH₂ClCOOH lactic acid, CH₂CH(OH)COOH nitrous acid, HNO₂ hydrofluoric acid, HF 10 2 7.6 X 10-3 1.4 x 10-3 8.4 X 10 4 4.3 X 10-4 3.5 x 10-4 pKa 0.52 0.70 0.77 1.81 2.00 2.12 2.85 3.08 3.37 3.45 Acid formic acid, HCOOH benzoic acid, C,H₂COOH acetic acid, CH,COOH carbonic acid, H₂CO3 hypochlorous acid, HCIO hypobromous acid, HBrO boric acid, B(OH)3¹ hydrocyanic acid, HCN phenol, C,H,OH hypoiodous acid, HIO K₂ 1.8 X 10 4 6.5 x 10-5 1.8 X 10-5 4.3 X 107 3.0 X 108 2.0 × 10-⁹ 7.2 X 10-10 4.9 X 10-10 1.3 X 10-10 2.3 X 10-11 pKa 3.75 4.19 4.75 6.37 7.53 8.69 9.14 9.31 9.89 10.64
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
CH32NH 1400 3...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Use data from Tables 6C.1 and 6C.2 to place the following acids in order of increasing strength: HCOOH, (CH 3 ) 3 NH + , N 2 H 5 + , HF. TABLE 6C.1 Acidity Constants at 25 C* K 3.0 X 10-1 2.0 X 10-...
-
a. Place the following acids in order of strength, starting with the strongest acid first. CH 3 CH 2 COOH CH 3 CCl 2 COOH CH 3 CHClCOOH b. Explain why ethanoic acid is a stronger acid than ethanol....
-
Use Paulings rules to place the following acids in order of increasing acid strength: HNO 2 , H 2 SO 4 , HBrO 3 , and HClO 4 in a nonlevelling solvent.
-
Metro Credit Union in Charlottetown, Prince Edward Island, loaned $90,000 to David Mann on a six-month, 8% note. Record the following for Metro Credit Union: a. Lending the money on March 6. b....
-
Effect of management evaluation criteria on EOQ model. Computers 4 U is an online company that sells computers to individual consumers. The annual demand for one model that will be shipped from the...
-
Forester and Cohen is a small accounting firm, managed by Joseph Cohen since the retirement in December of his partner Brad Forrester. Cohen and his 3 CPAs can together bill 640 hours per month. When...
-
Think of a time you were talking with someone and felt yourself getting angry. How did you handle it? What steps would you take to keep from getting angry with a customer who was being unreasonable?
-
The deepest point known in any of the earth's oceans is in the Marianas Trench, 10.92 k/D deep. (a) Assuming water is incompressible, what is the pressure at this depth? Use the density of seawater....
-
) 2345- ohao123_ w Quiz: Online Homework 4 + 3 -> C canvas.uw.edu/courses/1634621/quizzes/1823785/take M Gmail YouTube New folder useful Question 1 gs 24 2X R2.00 2X R.75 4X R2.50 NE 42nd St WB...
-
A combustion analysis of 1.200 g of an anhydrous sodium salt gave 0.942 g of CO 2 , 0.0964 g of H 2 O, and 0.246 g of Na. The molar mass of the salt is 112.02 g mol 1 . (a) What is the chemical...
-
Calculate the molar solubility of each substance in its respective solution: (a) Silver chloride in 0.20 m NaCl(aq); (b) Mercury(I) chloride in 0.150 m NaCl(aq); (c) Lead(II) chloride in 0.025 m CaCl...
-
Evaluate the definite integral. (x-2x) dx
-
Graph the DataIn this lab, "Position" is the dependent variable, so should go onthe y-axis and "Time", as theindependent variable goes on the x-axis. Part IIIn part I, you started the car at the...
-
answer all questions accordingly pls Cold Boot Switch/Router 1) ROM-POST (Power on Self-Test)-Several diagnostic programs that tests the hardware of the router 2) ROM-Bootstrap-check the...
-
tr P Final. Exam..Calculus. (3) Sec 5 (Calculus 3-Cl * .10 (3 ) 15tan-(-)dzdzdy- 3.77 O 6.77 O 127 O 15.7 O 18.72 J 97 O * .11 (2 ) Activate Win Go to Settings t
-
i need help with all these 42 CONCEPT APPLICATIONS Identify the parts of the circle shown below. 1. ZABC 3. AO 5. ZAOC 7. AB 9. Area between chord DE and the circle 10. Area bounded by CO, AO, and...
-
(b) Given the following data: name hair height weight lotion status Sarah blonde average light no sunburned Dana blonde tall average yes none Alex brown short average yes none Annie blonde short...
-
Robinsons, Inc., has employed a balanced scorecard for several months now. The firm distributes high fashion accessories to boutiques and high-end department stores. At these stores,...
-
What are some of the various ways to implement an awareness program?
-
Identify the reactants you would use to prepare each of the following azo dyes via an azo coupling reaction: (a) (b) (c) N- !i -NH2 S SO,H -NO2
-
Starting with benzene and isopropyl chloride, show how you would prepare the following compound: N- -NH2 O,N-
-
Draw the product obtained when the diazonium salt formed from aniline is treated with each of the following compounds: (a) Aniline (b) Phenol (c) Anisole (methoxybenzene)
-
A bank has a duration of 12 years for its assets and a duration of 18 years for its liabilities. Further, the leverage ratio (k) is 0.75. What will be the effect of rising interest rates on the...
-
Given that A = 2T-3j, and B = 51 +7j, find a unit vector parallel to A.
-
What is the major drawback of the upfront collection and documentation of requirements in the Waterfall methodology?

Study smarter with the SolutionInn App


