Use data from Tables 6C.1 and 6C.2 to place the following acids in order of increasing strength:
Question:
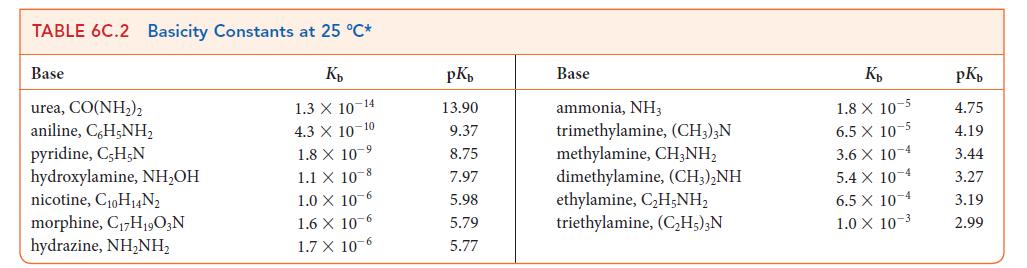
Use data from Tables 6C.1 and 6C.2 to place the following acids in order of increasing strength: HCOOH, (CH3)3NH+, N2H5+, HF.
Transcribed Image Text:
TABLE 6C.1 Acidity Constants at 25 C* K 3.0 X 10-1 2.0 X 10- 1.7 X 10-1 Acid trichloroacetic acid, CCI,COOH benzene sulfonic acid, C6H,SO3H iodic acid, HIO3 sulfurous acid, HSO3 chlorous acid, HClO phosphoric acid, H3PO4 chloroacetic acid, CHCICOOH lactic acid, CHCH(OH)COOH nitrous acid, HNO hydrofluoric acid, HF 1.5 X 10 2 1.0 X 10-2 7.6 X 10-3 1.4 X 10-3 8.4 X 10 4 4.3 X 10-4 3.5 X 10-4 pKa 0.52 0.70 0.77 1.81 2.00 2.12 2.85 3.08 3.37 3.45 Acid formic acid, HCOOH benzoic acid, C,HCOOH acetic acid, CH,COOH carbonic acid, HCO3 hypochlorous acid, HCIO hypobromous acid, HBrO boric acid, B(OH)3 hydrocyanic acid, HCN phenol, C,H,OH hypoiodous acid, HIO K 1.8 X 10-4 6.5 X 10-5 1.8 X 10-5 4.3 X 10-7 3.0 X 10 8 2.0 10-9 7.2 X 10-10 4.9 X 10-10 1.3 X 10-10 2.3 X 10-11 pKa 3.75 4.19 4.75 6.37 7.53 8.69 9.14 9.31 9.89 10.64
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Decreasing pKa will correspond t...View the full answer

Answered By

Divya Munir
I hold M.Sc and M.Phil degrees in mathematics from CCS University, India and also have a MS degree in information management from Asian institute of technology, Bangkok, Thailand. I have worked at a international school in Bangkok as a IT teacher. Presently, I am working from home as a online Math/Statistics tutor. I have more than 10 years of online tutoring experience. My students have always excelled in their studies.
4.90+
0 Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Use data from Tables 6C.1 and 6C.2 to place the following acids in order of increasing strength: HNO 2 , HClO 2 , +NH 3 OH, (CH 3 ) 2 NH 2 + . TABLE 6C.1 Acidity Constants at 25 C* K 3.0 X 10-1 2.0 X...
-
Use data from Tables 6C.1 and 6C.2 to place the following bases in order of increasing strength: C 10 H 14 N 2 (nicotine), ClO 2 , (CH 3 ) 3 N, HSO 3 . TABLE 6C.1 Acidity Constants at 25 C* K 3.0 X...
-
Use data from Tables 6C.1 and 6C.2 to place the following bases in order of increasing strength: F , NH 3 , CH 3 CO 2 , C 5 H 5 N (pyridine). TABLE 6C.1 Acidity Constants at 25 C* K 3.0 X 10-1 2.0...
-
Is land allowed to be depreciated? Why or why not?
-
New equipment purchase, income taxes Annas Bakery plans to purchase a new oven for its store. The oven has an estimated useful life of 4 years. The estimated pretax cash flows for the oven are as...
-
Rocky Mountain Tire Center sells 20,000 go-cart tires per year. The ordering cost for each order is $40, and the holding cost is 20% of the purchase price of the tires per year. The purchase price is...
-
You are vice president of sales for your company and are speaking with your sales managers from around the country. You have been asked by the CEO to prepare a 5-minute presentation on why time and...
-
During the 1980s, most of the worlds supply of lysine was produced by a Japanese company named Ajinomoto. Lysine is an essential amino acid that is an important livestock feed component. At this...
-
Strategic plans cannot always predict the future in times of uncertainty. Healthcare managers must take the time to reassess during these times to make improvements to guide future processes and...
-
The pH of several solutions was measured in a hospital laboratory; convert each of the following pH values into the molar concentration of H 3 O + ions: (a) 4.8 (the pH of a urine sample); (b) 0.7...
-
Calculate the molar solubility of each substance in its respective solution: (a) Silver iodide in 0.020 m NaI(aq); (b) Calcium carbonate in 2.3 * 10 4 m Na 2 CO 3 (aq); (c) Lead(II) fluoride in 0.21...
-
Lumberton Home Maintenance Company (LHMC) earned operating income of $6,000,000 on operating assets of $62,500,000 during 2018. The Tree Cutting Division earned $1,000,000 on operating assets of...
-
Yeats, Irons, and Rhoades (1975) found that annual deposit growth for 48 commercial banks can be modeled by the following function: D_(t+1)/D_t=1.172-0.125t^-1+1.135t^-2 where D is the year-end...
-
2. Graph y(x) = x. Express the domain and range for this function in interval notation.
-
Discuss the concepts of resolution and aliasing in DoE (use resolution IV as an example). b) 3 marks Which type of control chart is most appropriate to monitor the following situation? i) Bore...
-
Consider the model problem below, find A and R matrices, and b and r vectors. Then find the solution: -(xu'(x))' = 4x x EI = [0, 3] xu' (0)=0; u(L)=5; ko=0; k=1 u(0)=5; 9L=5; 90=1;
-
Q1 Hadamard's inequality (i) Let A = (a lan) E Rnxn. Let A = QR = Q(r|rn) be a QR factorization of A. - First, show that det(A)| II ri||2. Then, using the previous part, show that det(A)| II1...
-
Considerable research has been done on the relation between a firms earning stream and the market price for its stock. However, earnings explain just a small fraction of the variation in market...
-
What did Lennox gain by integrating their WMS, TMS, and labor management systems?
-
Propose an efficient synthesis for each of the following transformations. (a) (b) (c) (d) OEt EtO
-
The product of an aldol condensation is an α,β unsaturated ketone which is capable of undergoing hydrogenation to yield a saturated ketone. Using this technique, identify...
-
Identify the reagents you would use to convert cyclohexanone into each of the following compounds. (a) (b) (c) (d) (e) (f) (g)
-
Presented below is the trial balance of scott butler corporation at December 3 1 , 2 0 1 4 . Cash $ 1 9 7 , 0 0 0 Sales Revenue $ 8 , 1 0 0 , 0 0 0 Debt Investments ( trading ) ( at cost , $ 1 4 5 ,...
-
How does the average power due to the tension for the time interval from 0 to = 8 s compare to its instantaneous power at t = 8 s?
-
In a class of 50 students, a teacher surveyed the students and collected the following data: 24 play basketball, 27 play volleyball, and 29 play badminton. .18 play both basketball and volleyball. 16...

Study smarter with the SolutionInn App


