Use the phase diagram for helium in Exercise 5B.3 (a) To describe the phases in equilibrium at
Question:
Use the phase diagram for helium in Exercise 5B.3
(a) To describe the phases in equilibrium at each of helium’s two triple points;
(b) To decide which liquid phase is the more dense, helium-I or helium-II.
Exercise 5B.3
The phase diagram for helium is shown here.
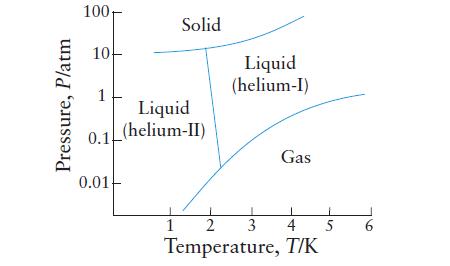
Transcribed Image Text:
Pressure, P/atm 100, 10 1 Solid Liquid 0.1(helium-II) 0.01 Liquid (helium-I) 1 3 Gas L 1 2 4 Temperature, T/K 5 6
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a Lower pressure triple point liquid ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Use the accompanying phase diagram for sulfur to answer the following questions. (The phase diagram is not to scale.) a. How many triple points are in the phase diagram? b. What phases are in...
-
Phase diagrams for materials that have allotropes can be more complicated than those shown in the chapter. Use the phase diagram for carbon given here to answer the following questions. (a) How many...
-
The phase diagram for SO2 is shown here. (a) What does this diagram tell you about the enthalpy change in the reaction SO2(I) SO2(g)? (b) Calculate the equilibrium constant for this reaction at 100...
-
In Problems 25 54, solve each system. Use any method you wish. 2xxy + y = 8 xy = 4
-
A stream containing 7,000 kmol/h of water and 3,000 parts per million (ppm) by weight of ammonia at 350 K and 1 bar is to be processed to remove 90% of the ammonia. What type of separation operation...
-
On August 18, 1993, a fare war erupted. To initiate its new service between Cleveland and Baltimore, Southwest announced a $49 fare (a sizable reduction from the then standard rate of $300). Its...
-
Two plates are at temperatures of \(T_{1}\) and \(T_{2}\) and a chemical reaction is producing heat at a constant rate within the system. Derive a model to predict the temperature distribution within...
-
Following is selected information from the balance sheet for Flaps Inc. Solve for the missing amounts for each of the five years. Year 2016 2017 2018 2019 2020 Total liabilities and stockholders...
-
Part 1: Email Format Give an email to your instructor to ask for a meeting. Use your instructor's full name in the TO: line Use the format, structure and design Course Competencies from the course to...
-
The unadjusted trial balance of Mesa Inc., at the company's year end of December 31 follows: Additional information and adjustment data: 1. The 12-month insurance policy was purchased and was...
-
In a gas-phase equilibrium mixture of PCl 5 , PCl 3 , and Cl 2 at 500. K, PPCl 5 = 1.18 bar, PCl 2 = 5.43 bar. What is the partial pressure of PCl 3 , given that K = 25 for the reaction PCl 5 (g) PCl...
-
The following flasks show the dissociation of a diatomic molecule, X 2 , over time. (a) Which flask represents the point in time at which the reaction has reached equilibrium? (b) What percentage of...
-
What would a long bone look like at the end of adolescence if bone remodeling did not occur?
-
Based on what the IC considers important for equity-related strategies, which strategy should Mukilteo most likely avoid? A. Long/short equity B. Equity market neutral C. Dedicated short selling and...
-
Which of the IC members statements regarding hedge fund strategies is incorrect? A. Statement 1 B. Statement 2 C. Statement 3 Snohomish Mukilteo is a portfolio analyst for the Puyallup-Wenatchee...
-
Which of the following notes about the DoGood Fund is correct? A. Only Note 2 B. Only Note 3 C. Both Note 2 and Note 3 Three years ago, the Albright Investment Management Company (Albright) added...
-
The government bond strategy that Mukilteo considers is best described as a: A. carry trade. B. yield curve trade. C. long/short credit trade. Snohomish Mukilteo is a portfolio analyst for the...
-
Which of Freys statements about securities lending and covered call writing is correct? A. Only Statement 1 B. Only Statement 2 C. Both Statement 1 and Statement 2 Three years ago, the Albright...
-
Calculate the annual difference between the cash flow and the deductibility for tax purposes of the purchase of a $20,000 truck. The truck is depreciated using the half-year convention and the 200%...
-
What are the two methods used to translate financial statements and how does the functional currency play a role in determining which method is used?
-
A sample of propane (C 3 H 8 ) is placed in a closed vessel together with an amount of O 2 that is 2.15 times the amount needed to completely oxidize the propane to CO 2 and H 2 O at constant...
-
Propose a structure for a compound with molecular formula C 10 H 14 O that exhibits the following 1 H NMR spectrum. Proton NMR Chemical shift (ppm)
-
A van der Waals gas undergoes an isothermal reversible expansion under conditions such that z < 1. Is the work done more or less than if the gas followed the ideal gas law?
-
Write a polynomial f(x) that meets the given conditions. Answers may vary. Degree 3 polynomial with zeros 1, -5, and 3. f(x) =
-
For the real-valued functions g(x) = x +3 and h(x)=x+2, find the composition gh and specify its domain using interval notation.
-
For the straight line defined by the points (4,59) and (6,83), determine the slope (m) and y-intercept (b). Do not round the answers. m = b=

Study smarter with the SolutionInn App


