Using Fig. 12.29, list elements (ignore the lanthanide and actinides) that have ground-state electron configurations that differ
Question:
Using Fig. 12.29, list elements (ignore the lanthanide and actinides) that have ground-state electron configurations that differ from those we would expect from their positions in the periodic table.
Fig. 12.29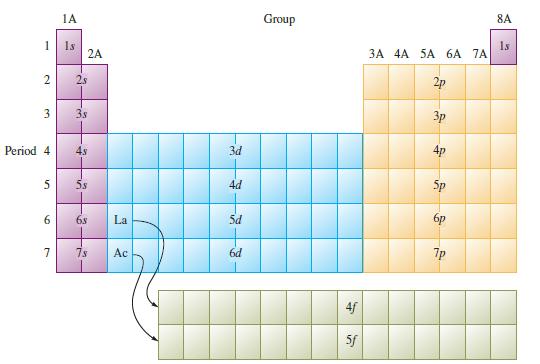
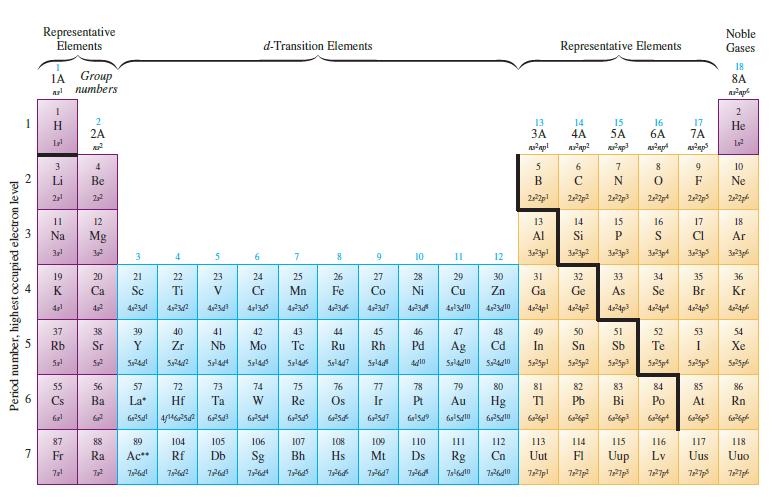
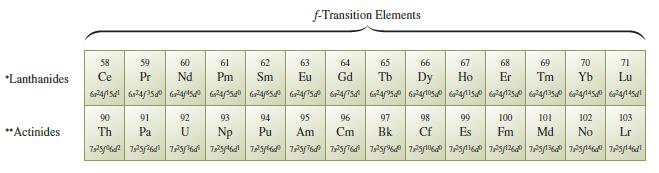
Transcribed Image Text:
1 2 3 Period 4 5 6 7 1A 1s -3-3- 2A 2s 38 4s 58 68 La 7s Ac 3d 4d 5d 6d Group 4f 5f 3A 4A 5A 6A 7A 2p 3p 4p 5p 7p 8A 1s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Total 14 elements except lanthanide and actinide sh...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
On the basis of their positions in the periodic table, select the atom with the larger atomic radius in each of the following pairs: (a) Na, Cs; (b) Be, Ba; (c) N, Sb; (d) F, Br; (e) Ne, Xe.
-
Based on their positions in the periodic table, predict which atom of the following pairs will have the smaller first ionization energy: (a) Cl, Ar; (b) Be, Ca; (c) K, Co; (d) S, Ge; (e) Sn, Te.
-
We have considered helium configurations in which only one electron is excited. Get a rough estimate of the energy of the 2s2 configuration from Eq. (9.48). Compare this with the ground-state energy...
-
For the increasing marginal-extraction-cost model of the allocation of a depletable resource, how would the ultimate cumulative amount taken out of the ground be affected by (a) An increase in the...
-
Find the z-score that has 78.5% of the distributions area to its right.
-
Based on the dangling-else discussion in Exercise 4.27, modify the following code to produce the output shown. Use proper indentation techniques. You must not make any additional changes other than...
-
What do you like best about working at this company?
-
Atwood Company has an opportunity to produce and sell a revolutionary new smoke detector for homes. To determine whether this would be a profitable venture, the company has gathered the following...
-
At the Natural History Museum, tickets cost $10 for adults and $6 for kids under 12. What would be the total cost for 12 adult tickets and 18 kids tickets? What would be the total cost for a adult...
-
Due to the integrated nature of their capital markets, investors in both the United States and the U.K. require the same real interest rate, 2.5 percent, on their lending. There is a consensus in...
-
Using the element phosphorus as an example, write equations for the processes in which the energy change will correspond to the ionization energy and to the electron affinity.
-
Which of the following is the best reducing agent: F 2 , H + , Na, Na + , or F 2 ? Explain. Order as many of these species as possible from the best to the worst oxidizing agent. Why cant you order...
-
Does sampling risk always exist in both statistical and non-statistical sampling? Explain your response.
-
On the first day of the fiscal year, Rainbow Company issues a $692200, 6%, 5-year bond that pays semiannual interest for a cash price of 95% of the face value of the bond. What is the discount amount?
-
1. Suppose that a BMW costs 45,000 in Germany and that the current USD/EUR exchange rate is .80. a) Calculate the dollar price of the BMW. b) Now suppose the euro appreciates relative to the dollar...
-
Under a divorce agreement executed in 2017 Roberta receives $2,000 in monthly alimony from her former husband and $3,000 in child support payments. How much of her annual payments received in 2023...
-
This assignment requires you to complete a number of typical tasks that you would normally use JavaScript to solve. Each task is typical situation that requires you to apply specific JavaScript...
-
JCR Co. reported $770,000 in net income and its weighted average shares outstanding for the year is 100,000 shares. In prior years it sold $1,000,000 of 8% long-term convertible bonds at par which...
-
In forecasting a monthly time series over a five-year period from January 2006 to December 2010, the exponential trend forecasting equation for January is Take the antilog of the appropriate...
-
Vectors are drawn from the center of a regular n-sided polygon in the plane to the vertices of the polygon. Show that the sum of the vectors is zero.
-
Consider the reaction 4PH3(g) P4(g) + 6H2(g) If, in a certain experiment, over a specific time period, 0.0048 mole of PH3 is consumed in a 2.0- L container during each second of the reaction, what...
-
In the Haber process for the production of ammonia, N2(g) + 3H2(g) 2NH3(g) What is the relationship between the rate of production of ammonia and the rate of consumption of hydrogen?
-
What are the units for each of the following if concentrations are expressed in moles per liter and time in seconds? a. Rate of a chemical reaction b. Rate constant for a zero-order rate law c. Rate...
-
Why groupthink is not good for the group? Write with example. What are the differences between Divergent and Convergent thinking?
-
Successful management communication can make a massive difference in realizing change, and requires that managers, as leaders, reflect on how they communicate with others and how they frame change to...
-
1. Explain the meaning and what things need to be considered in "Acquiring IS & Application" 2. Give examples of cases of implementation in industry in Indonesia and their linkages with existing...

Study smarter with the SolutionInn App


