Which of the following processes are exothermic? a. N(g) 2N(g) b. HO(l) HO(s) c. Cl(g)
Question:
Which of the following processes are exothermic?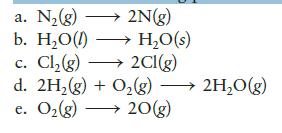
Transcribed Image Text:
a. N₂(g) 2N(g) b. H₂O(l) → H₂O(s) c. Cl₂(g) →→→ 2Cl(g) d. 2H₂(g) + O₂(g) →→→→ 2H₂O(g) e. O₂(g) → 20(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
c Cl2g 2Clg d 2H2g O2g 2HOg Exothermic processes are chemical processes that involve the release of ...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following processes are allowed in electromagnetic interactions and which are allowed in weak interactions via the exchange of a single W or Z 0 ? (a) K + 0 + e + + e , (b) K + + +...
-
Which of the following processes will likely result in a precipitation reaction?
-
Which of the following processes has a AS 0 A) 2H, () O, () 2H,0 (g) B) 2NO2 g) Ba (aq) 2F (aq) 2HgO (s) OB
-
Let F = [F] lb. a) Determine the force Fp acting at roller D. b) Determine the force FE acting at pin E. F 4 ft B 600 lb. ft -3 ft C -3 ft OD -2 ft- E
-
Suppose that a random sample of n observations is drawn from a t distribution with > 2 degrees of freedom. Show that the asymptotic distributions of both the sample mean n and the sample median are...
-
Use AnswerConnect to answer the following questions: a. What does IRS Notice 2016-55 discuss? On what date was it issued? b. Who is the author of Canceled S Corporation Debt and Deemed NOLs in...
-
What are the two categories of data mining and knowledge discovery software?
-
Consider the following activity information and the constraint that the project must be completed in 16 weeks. In addition, activities c, f, h, and i may be crashed as follows. Assume partial...
-
A seal dives below the ice in search of fish. Its path can be modelled by where d is the depth in decameters and t the time in minutes. Use the equation above to answer questions about the seal's...
-
Compute and compare the accounts receivable turnover ratios for coca-cola and Walmart. Indicate all numbers you used to calculate the ratio. What constitutes a reasonable account receivable turnover...
-
In a coffee-cup calorimeter, 150.0 mL of 0.50 M HCl is added to 50.0 mL of 1.00 M NaOH to make 200.0 g solution at an initial temperature of 48.28C. If the enthalpy of neutralization for the reaction...
-
In which of the following systems is(are) work done by the surroundings on the system? Assume pressure and temperature are constant. a. 2SO(g) + O(g) 2SO3(g) b. CO (s) CO(g) c. 4NH3(g) + 70(g) d....
-
In each of the following cases, determine whether the sample size n is large enough to say that the sampling distribution of p is a normal distribution. a. p = .4. n = 100 b. p = .1. n = 10 c. p =...
-
How to Proceed write an assignment of one thousand two hundred Guidelines on word count for each section are provided below. Identify a past or current personal or professional initiative classified...
-
Which of the 12 Principles best addresses the project development approach of addressing the unique nature of each project in order to deliver value most effectively? Recognize, evaluate, and respond...
-
Use the following information to answer the next three questions. A student performed an experiment where the motion of a basketball was studied as it rolled along an inclined ramp. Direction of...
-
Write in LISP the expression equivalent to the following Java expression. (123) && ! (3 <4) Write one line and add a whitespace between each word, number or symbol in your answer. As done in the...
-
TASK: Write own word reflection on the things that you learned in Hospitality Supervisor Certificate. After taking the course, do you think that you are now ready to be a good leader in the future?...
-
The maximum flow rate of standard shower heads is about 3.5 gpm (13.3 L/min) and can be reduced to 2.75 gpm (10.5 L/min) by switching to low-flow shower heads that are equipped with flow controllers....
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
Consider the following galvanic cell: Calculate the concentrations of Ag+(aq) and Ni2+(aq) once the cell is dead. Ni 1.0 MNi2+ 10 M Ag
-
A galvanic cell is based on the following half reactions: Fe2+ + 2e Fe(s) o = 20.440 V 2H+ + 2e H2(g) o = 0.000 V In this cell the iron compartment contains an iron electrode and [Fe2+] = 1.00 103...
-
You have a concentration cell with Cu electrodes and [Cu2+] = 1.00 M (right side) and 1.0 1024 M (left side). a. Calculate the potential for this cell at 25oC. b. The Cu21 ion reacts with NH3 to...
-
This was the first time in history that a cyberattack was synchronized with a military campaign. A. World War III B. Ukrainian Revolution C. Russo-Georgian War D. Civil War
-
Based off the below table write a query to get the quantities of the sold products on Wednesday in the category Footwear provided by vendor Pacific Gear within the Tristate region between the 1st and...
-
the process of identifying actors, use cases, creating a table with descriptions, listing flow of events, and sketching a use-case diagram for a Course Registration System involves the following...

Study smarter with the SolutionInn App


