Write the expression for K for each of the following reactions: (a) 2 CH4 (g) + O(g)
Question:
Write the expression for K for each of the following reactions: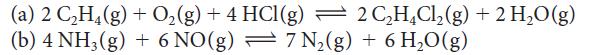
Transcribed Image Text:
(a) 2 C₂H4 (g) + O₂(g) +4 HCl(g) — 2 C₂H4Cl₂(g) + 2 H₂O(g) (b) 4 NH3(g) + 6 NO(g) = 7 N₂(g) + 6 H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a K b K P...View the full answer

Answered By

Pharashram rai
i am highly creative, resourceful and dedicated freelancer an excellent record of successful classroom presentations and writing . I have more than 4 years experience in tutoring students especially by using my note making strategies and engineering field . Especially adept at teaching methods of maths and writing , and flexible teaching style with the willingness to work beyond the call of duty. Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with .
4.80+
65+ Reviews
270+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Identify the alkene product in each of the following Wittig reactions
-
Write the expression for Kc for the following reactions. In each case indicate whether the reaction is homogeneous or heterogeneous.
-
Give the expression for K for each of the following reactions. a. b. c. CaCO (s)CaO(s) CO2(g) Pbl2(s) H (a) HCO (aq)H2O) CO2(g)
-
Shown here is a list published by Electronics Weekly.com of the top five semiconductor companies in the United States by revenue ($ billions). a. Construct a bar chart to display these data. b....
-
A conducting sphere of radius R1 is given a free charge Q. The sphere is surrounded by an uncharged concentric spherical dielectric shell having an inner radius R1, an outer radius R2, and a...
-
In many cases, organizations use teams as a part of their high-performance work systems. Could such systems be useful in organizations that do not use teams? What special concerns might you have to...
-
Water is boiled in a polished stainless steel pot with a \(3 \mathrm{~kW}\) heater. The efficiency of the heater is \(60 \%\), i.e., only \(60 \%\) of the heat is transferred to the water. Find the...
-
Q1. The following transactions occurred during Year 1, the first year of business, for Nancy Nanny Child Care. Record each transaction in proper journal entry format below using debits and credits....
-
What strategies can leaders employ to cultivate a culture of collaboration that transcends geographical boundaries and time zones, especially within multinational corporations operating in diverse...
-
Harry Jamison, CPA, is planning to set up a business to prepare tax returns. Harry is the only person in the business, at least for now. Can he delegate any work? Should he? Explain.
-
The phase diagram for helium is shown here. (a) What is the maximum temperature at which superfluid helium-II can exist? (b) What is the minimum pressure at which solid helium can exist? (c) What is...
-
Complete the following statements about the effect of intermolecular forces on the physical properties of a substance. (a) The higher the boiling point of a liquid, the (stronger, weaker) are its...
-
Clarke Inc. operates the Patio Furniture Division as a profit center. Operating data for this division for the year ended December 31, 2022, are as shown below. In addition, Clarke incurs $180,000 of...
-
What is the value of a derivative that pays off \($100\) in 6 months if an index is greater than 1,000 and zero otherwise? Assume that the current level of the index is 960, the risk-free rate is 8%...
-
Aerotron Electronics is considering purchasing a water filtration system to assist in circuit board manufacturing. The system costs \(\$ 40,000\). It has an expected life of 7 years at which time its...
-
Two incinerators are being considered by a waste management company. Design A has an initial cost of \($2\),500,000, has annual operating and maintenance costs of \($800\),000, and requires overhauls...
-
When writing a cover letter, its relatively easy to identify key words from a particular employers help wanted ad or website. When you write a LinkedIn profile, you are casting a wider net. What are...
-
Repeat Problem 9 assuming that the cost of capital has increased due to weak market conditions and MARR is now 6 percent/year. Data from problem 9 Bailey, Inc., is considering buying a new gang punch...
-
Your construction company recently bid against ABC Construction Company. Your construction costs were $265,815 and you added an 11% profit and overhead markup for a total bid of $295,055. ABCs bid...
-
On January 1, 2017, McIlroy, Inc., acquired a 60 percent interest in the common stock of Stinson, Inc., for $340,200. Stinson's book value on that date consisted of common stock of $100,000 and...
-
For each of the following reactions, predict the product and draw the mechanism of its formation. a. b. c. d. e. f. 1) PhMgBr 2) H20 Me 1) NaCN 2) H20 *Me
-
For values of z near one, it is a good approximation to write z(P) = 1 + (z/P) T . P. If z = 1.00104 at 298 K and 1 bar, and the Boyle temperature of the gas is 155 K, calculate the values of a, b,...
-
A 1.75 mole sample of an ideal gas for which P = 2.50 bar and T = 335 K is expanded adiabatically against an external pressure of 0.225 bar until the final pressure is 0.225 bar. Calculate the final...
-
A conducting wire of length 2.61 m has a current of 14.4 A passing through it. The wire is placed in a magnetic field of strength 1.58 T. If the angle between the wire and the magnetic field is 45.3,...
-
A solenoid is created using a wire wrapped in a coil of radius 2 cm and length 20 cm. If the coil has 175 turns and carries a current of 10 A, what is the magnitude of the magnetic field inside the...
-
An engine produces 28 kW of power while operating with 4.5% exhaust oxygen (wet measurement), and C1H2.1O0.2 fuel. The reading from the mass air flow (MAF) meter is 26 g/s. Calculate the brake...

Study smarter with the SolutionInn App


