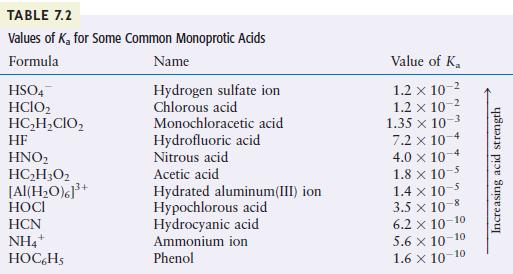
You may need Table 7.2 to answer the following questions. a. Which is the stronger acid, (mathrm{H}_{2}
Question:
You may need Table 7.2 to answer the following questions.
a. Which is the stronger acid, \(\mathrm{H}_{2} \mathrm{SO}_{4}\) or \(\mathrm{H}_{2} \mathrm{O}\) ?
b. Which is the stronger acid, \(\mathrm{H}_{2} \mathrm{O}\) or \(\mathrm{HOCl}\) ?
c. Which is the stronger acid, \(\mathrm{NH}_{4}{ }^{+}\)or \(\mathrm{HC}_{2} \mathrm{H}_{2} \mathrm{ClO}_{2}\) ?
Transcribed Image Text:
TABLE 7.2 Values of K for Some Common Monoprotic Acids Formula Name Hydrogen sulfate ion Chlorous acid HSO4 HCIO HCHCIO HF HNO HCH3O2 [Al(HO)]+ HOCI HCN NH4+ HOC6H5 Monochloracetic acid Hydrofluoric acid Nitrous acid Acetic acid Hydrated aluminum(III) ion Hypochlorous acid Hydrocyanic acid Ammonium ion Phenol Value of K 1.2 x 10 1.2 x 10-2 1.35 x 10-3 7.2 x 10-4 4.0 x 10 1.8 x 10 1.4 x 10- 3.5 x 10-8 6.2 10-10 5.6 10-10 1.6 10-10 Increasing acid strength
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Based on the provided Table 72 which shows values of the acid dissociation constant K for some commo...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
You may need Table to answer the following questions. a. Which is the stronger acid, HCl or H2O? b. Which is the stronger acid, H2O or HNO2? c. Which is the stronger acid, HCN or HOC6H5? Table...
-
You may need Table to answer the following questions. a. Which is the stronger base, Cl2 or H2O? b. Which is the stronger base, H2O or NO2-? c. Which is the stronger base, CN2 or OC6H5-? Table...
-
Linda has d dollars in an account that pays 1.4% interest, compounded weekly. She withdraws w dollars. Express her first weeks interest algebraically.
-
A transparent diffraction grating of a quartz spectrograph is 25 mm wide and has 250 lines per millimeter. The focal length of an objective in whose focal plane a photographic plate is located is...
-
Using the mono alphabetic cipher in Figure 8.3, encode the message "This is an easy problem." Decode the message "rmij'u uamu xyj."
-
Plaintiff contracted to install a boiler for defendant. After plaintiff had installed and tested the boiler, but before final payment to plaintiff had been made, defendant took custody of the boiler...
-
Toshihiro Nakamura, manufacturing engineering section manager, is examining the prototype assembly process sheet (shown in Exhibit 8.15) for the newest subnotebook computer model. With every new...
-
The local theater company is choosing its next play. The company's members recently held a vote to decide which of three choices - Elektra, Tartuffe, and Hamlet - the company will perform. The...
-
You may need Table 7.2 to answer the following questions. a. Which is the stronger base, \(\mathrm{HSO}_{4}{ }^{-}\)or \(\mathrm{H}_{2} \mathrm{O}\) ? b. Which is the stronger base, \(\mathrm{H}_{2}...
-
Use Table 7.2 to order the following from the strongest to the weakest acid. \[\mathrm{HClO}_{2}, \mathrm{H}_{2} \mathrm{O}, \quad \mathrm{NH}_{4}^{+}, \quad \mathrm{HClO}_{4}\] TABLE 7.2 Values of K...
-
Solve the initial value problem by a power series. Graph the partial sums of the powers up to and including x 5 . Find the value of the sum s (5 digits) at x 1 . (x - 2)y' = xy, y(0) = 4, x 1 = 2
-
You will play the role of a customer in a restaurant. Choose a restaurant that you have never eaten at before and that has wait-staff to serve you at your table ( not a self-serve, fast food or...
-
Critically explain the political factors affecting Air canada. please give detailed explanation and also add references
-
1. Identify two firms in two different industries that you believe exhibit a nimble nature in their operations. 2. What specific evidence leads you to believe these firms are nimble, especially in...
-
Project selection sets an organization on the path to use of its limited resources. Without a sound project selection method, an organization may not be as efficient as they otherwise could be....
-
December 31, 2030 Accounts Debit Credit Cash Accounts Receivable 105,800 14,600 Allowance for Bad Debts Inventory Prepaid Advertising Supplies Accounts Payable Unearned Revenue Long-Term Note Payable...
-
In 1985, Bruce Byrne, with his sons Scott and Gordon, opened Lone Star R.V. Sales, Inc., a motor home dealership in Houston, Texas. In 1994, Lone Star became a franchised dealer for Winnebago...
-
The May 2014 revenue and cost information for Houston Outfitters, Inc. follow: Sales Revenue (at standard).............. $ 540,000 Cost of Goods Sold (at standard) ..........341,000 Direct Materials...
-
(a) Using the particle-in-the-box model for the hydrogen atom and treating the atom as an electron in a one-dimensional box of length 150. pm, predict the wavelength of radiation emitted when the...
-
At the time that J . J. Thomson conducted his experiments on cathode rays, the nature of the electron was in doubt. Some considered it to be a form of radiation, like light; others believed the...
-
Which of the following increase when an electron in a lithium atom undergoes a transition from the 1s-orbital to a 2p-orbital? (a) Energy of the electron. (b) Value of n. (c) Value of l. (d) Radius...
-
Accounts that have more of this characteristic are considered closer to cash and appear near the top of U.S. balance sheets. Name the characteristic (one word)
-
According to the Strategic Profit Model, which of these will lead to an increase in Return on Assets if all other values stay the same? Group of answer choices All of these answers A decrease in...
-
Flying Fish Brewing Company Review the case and based on your chapter learning and topics covered so far, answer the following questions. Flying Fish Brewing Company opens in new window Q1. Of the...

Study smarter with the SolutionInn App


