Gaseous reactant A diffuses through a gas film and reacts on the surface of a solid according
Question:
Gaseous reactant A diffuses through a gas film and reacts on the surface of a solid according to a reversible first-order rate,
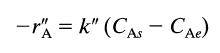
where CA, is the concentration of A in equilibrium with the solid surface. Develop an expression for the rate of reaction of A accounting for both the mass transfer and reaction steps.
Transcribed Image Text:
-ra= k" (CAS- CAe) A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
In order to develop an expression that accounts for both the mass transfer and reaction steps we need to consider the two different rates 1 The mass t...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
In chemical vapor deposition (CVD), a semiconducting or insulating solid material is formed in a reaction between a gaseous species and a species adsorbed on the surface of silicon wafers (disks...
-
The Corporate Average Fuel Economy (CAFE) regulations were put into law by Congress in 1975 to promote the sale of fuel-efficient automobiles and light trucks. The law requires automakers to boost...
-
Discuss the advantages and disadvantages of one of the financial innovations you select. (For example, sweep accounts, junk bond, smart card etcSelect one of them, then explain briefly and afterwards...
-
An article in Sociological Methods & Research (May 2001) analyzed the data presented in the accompanying table. A sample of 262 Kansas pig farmers was classified according to their education level...
-
What problems do analysts often encounter when they try to implement an ERP package?
-
Raymond Inc. manufactures desks. The following data was given for production in February: units, beginning work in process 0 units started 200 units completed 150 units, ending work in process (40%...
-
The online review service Yelp helps millions of consumers find the goods and services they seek. To help consumers make more-informed choices, Yelp includes over 120 million reviews. The file yelp...
-
Update the trial balance for Kylers Moving Co. (Figure) for January 31, 201X. Adjustment Data to Update Trial Balance a. Insurance expired, $550. b. Moving supplies on hand, $700. c. Depreciation on...
-
Argue that international space law is adequate and will explain why, using examples. Argue that current space law is not adequate and explain why, using examples. Cite source
-
In slurry reactors, pure reactant gas is bubbled through liquid containing suspended catalyst particles. Let us view these kinetics in terms of the film theory, as shown in Fig. P17.3. Thus, to reach...
-
Aqueous A (C AO = 50 mol/m 3 ) with physical properties close to water (p =1000 kg/m 3 , D -9 = m 2 /s) reacts by a second-order reaction (k = m 3 /mol s) as it flows at 10 mm/s through a tubular...
-
Suppose that one of the countries discussed in question (1) imposed a tariff, or a tax on imports, which your firm must then add to the cost of exporting to that country. The tariff does not apply;...
-
As preparation for the final research paper, formulate a theory about the correlation between a measurable dependent variable (the effect) and measurable independent variables (the causes). The topic...
-
Consider the following class Method Trace: What does the following code print out? public class MethodTrace { public MethodTrace ( ) { } public void sayCheese ( ) { } System.out.print ("Cheese");...
-
You are to conduct an interview with General Eugene Irwin. You are to assume the role of his character and answer the following three questions. Movie: The Last Castle 2001 1. Why do you believe you...
-
Evacuation of staff and customers Security management of cash, documents, equipment, keys or people Handling chemicals and hazardous substances Hazard identification and reporting Incident and...
-
Question 3. Suppose an MLB pitcher has two options for each pitch, throw fastball or throw curveball, and the batter has two options for each pitch, anticipate fastball or anticipate curveball. This...
-
The following calendar year-end information is taken from the December 31, 2015, adjusted trial balance and other records of Leone Company. Advertising expense . . . . . . . . . . . . . . . . . . . ....
-
Anne is employed by Bradley Contracting Company. Bradley has a $1.3 million contract to build a small group of outbuildings in a national park. Anne alleges that Bradley Contracting has discriminated...
-
Go to the five LearnChemE screencasts link for Chapter 5 (http://www.umich.edu/~elements/6e/05chap/learn-cheme-videos.html). 1. In the screencast of the PBR with pressure drop, is there a problem...
-
How would you modify Table 6-2 for a. A constant-volume gas-phase reaction, and b. A variable-volume gas-phase reaction? Table 6-2 1. Mole balances: BR dNAdt=rAV dNBdt=rBV dNCdt=rCV dNDdt=rDV PFR PBR...
-
(a) Without referring back, make a list of the most important items you learned in this chapter. (b) Overall, what do you believe were the three major purposes of the chapter?
-
My question is how should this look. Below this is my chart but not sure if I am corrrectly? The purpose of this assignment is to practice on the creation of a Relational Data Model from an Entity...
-
You will need $100 in 10 years. What is the amount you need to invest today in an account that earns 6% annual interest?
-
You have $500 in an account that earns 2.5% annual interest. How many years will it take for the amount to grow to $1,000?

Study smarter with the SolutionInn App


