Pure gaseous A at about 3 atm and 30C (120 mmollliter) is fed into a 1-liter mixed
Question:
Pure gaseous A at about 3 atm and 30°C (120 mmollliter) is fed into a 1-liter mixed flow reactor at various flow rates. There it decomposes, and the exit concentration of A is measured for each flow rate. From the following data find a rate equation to represent the kinetics of the decomposition of A. Assume that reactant A alone affects the rate.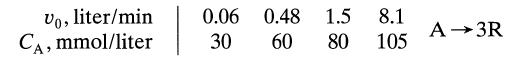
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





