The following data on an irreversible reaction are obtained with decaying catalyst in a batch reactor (batch-solids,
Question:
The following data on an irreversible reaction are obtained with decaying catalyst in a batch reactor (batch-solids, batch-fluid) What can you say about the kinetics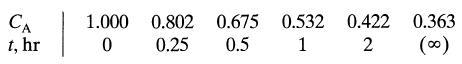
Transcribed Image Text:
CA t, hr 1 1.000 0 0.802 0.25 0.675 0.532 0.422 0.5 1 2 0.363 (00)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 20% (5 reviews)
The data you have provided shows the concentration of reactant A CA over time for an irreversible re...View the full answer

Answered By

Sagar Kumar
I am Mechanical Engineer with CGPA of 3.98 out of 4.00 from Pakistan. I went to Government Boys Degree College, Sehwan for high school studies.
I appeared in NUST Entrance Exam for admission in university and ranked #516. My mathematics are excellent and I have participated in many math competitions and also won many of them. Recently, I participated in International Youth Math Challenge and was awarded with Gold Honor. Now, I am also an ambassador at International Youth Math Challenge,
I have been teaching when I was in 9th class class year 2012. I have taught students from 6th class to university level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The reversible catalytic reaction proceeds with decaying catalyst in a batch reactor (batch-solids, batchfluid). What can you say of the kinetics of reaction and deactivation from the following data:...
-
The kinetics of a particular catalytic reaction A R are studied at temperature T in a basket reactor (batch-solids and mixed flow of gas) in which the gas composition is kept unchanged, despite...
-
The kinetics of a particular catalytic reaction A R are studied at temperature T in a basket reactor (batch-solids and mixed flow of gas) in which the gas composition is kept unchanged, despite...
-
4. Papo and Pepe are two barbers from a small barbershop. Theyhave their two court chairs plus two waiting chairs. The followingresults were found: P0 = 1/16 P1 = 4/16 P2 = 6/16 P3 = 4/16 a. What is...
-
The following transactions were completed by Prairie Renaissance Inc., whose fiscal year is the calendar year: 2005 July 1. Issued $8,000,000 of 5-year, 8% callable bonds dated July 1, 2005, at an...
-
Journalize and post the preceding transactions (for Exercise 15A-2) using a two-column journal and T accounts. 201X Nov. 5 Purchased on account 13 units at a cost of $10 each. (Inventory on hand...
-
What is the most valuable commodity from a trending program that must be communicated to management of a given facility?
-
Suppose you hold two series of options, both NQOs. Because of a big promotion, you expect your tax rate to increase from a current 31% to 39.6%. The current stock price is $70. The first set of...
-
what is the youngs modulus of a strand of spaghetti with weight 1 gram and length of 2 5 cm
-
A recycle reactor with very high recycle ratio is used to study the kinetics of a particular irreversible catalytic reaction, A R. For a constant flow rate of feed (' = 2 kg sec/liter) the...
-
In an automobile's catalytic converter, CO and hydrocarbons present in the exhaust gases are oxidized. Unfortunately the effectiveness of these units decreases with use. The phenomenon was studied by...
-
On March 20, Shelton Fireworks purchases on account $10,000 of fireworks. On March 25, Shelton returns $1,000 of the merchandise to the vendor. Shelton then pays the remaining bill, less a 3%...
-
Discuss the concepts of resolution and aliasing in DoE (use resolution IV as an example). b) 3 marks Which type of control chart is most appropriate to monitor the following situation? i) Bore...
-
In his early 20s, Zach purchased a life insurance policy on his life worth $1,000,000. He named his estate as the beneficiary. Every year he paid the annual premium of $10,000 until his untimely...
-
Skylar is a 27-year-old student. She goes to class full-time and tends bar at nights and on weekends, earning about $30,000 annually. Skylar's boss has told her that the bar is not doing well and may...
-
A company's 2022 net sales are $80,187. The previous fiscal year, net sales were $76,118. What would the sales be for a 24 month pro forma projection? Show your work and explain how you arrived at...
-
Evaluate each expression, using the values given in the following table X -3 -2 -10123 -183-15 f(x) 0 g(x) 2 6 7 -1 0 N -1 -8 (a) (go f)(-2) = (b) (fog)(1) = = (c) (fof)(3) =
-
Who regulates the regulators? Is it a problem?
-
Interview managers at three companies in your area about their use of ERP. How have their experiences been similar? What accounts for the similarities and differences?
-
The elementary gas-phase reaction A + B 2C in Problem P11-8B is now continued and carried out in packed-bed reactor. The entering molar flow rates are F A0 = 5 mol/s, F B0 = 2F A0 , and F I = 2F A0...
-
Use the reaction data in Problems P11-4A and P12-7B for the case when heat is removed by a heat exchanger jacketing the reactor. The flow rate of coolant through the jacket is sufficiently high that...
-
Derive the energy balance for a packed-bed membrane reactor. Apply the balance to the reaction in Problem P11-5A A B + C for the case when it is reversible with KC = 1.0 mol/dm 3 at 300 K. Species C...
-
The formula p = 4 x 2 + 1 2 . 7 models the projected population of a particular town, P , in thousands x years after 2 0 1 0 . In what year ( approximately ) do we expect the population to be 3 0 , 0...
-
16. Add or subtract the radical expressions as indicated, if possible. 92+162 - 318
-
The manager at a coffee stand keeps track of the number of cups of coffee and cups of tea sold each day and the total money received. On Saturday, a total of 4 3 cups were sold, and the money...

Study smarter with the SolutionInn App


