A city of 100,000 people uses approximately 1.0 * 10 11 kJ of energy per day. Suppose
Question:
A city of 100,000 people uses approximately 1.0 * 1011 kJ of energy per day. Suppose all of that energy comes from the combustion of liquid octane (C8H18) to form gaseous water and gaseous carbon dioxide. Use standard enthalpies of formation to calculate ΔH°rxn for the combustion of octane and then determine the number of kilograms of octane necessary to provide this amount of energy.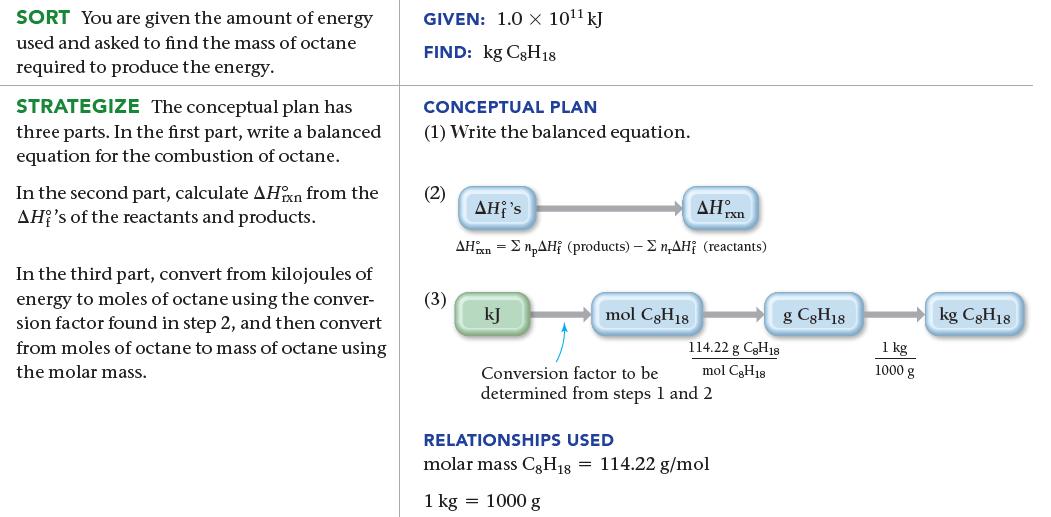
Transcribed Image Text:
SORT You are given the amount of energy used and asked to find the mass of octane required to produce the energy. STRATEGIZE The conceptual plan has three parts. In the first part, write a balanced equation for the combustion of octane. In the second part, calculate AHin from the AH's of the reactants and products. In the third part, convert from kilojoules of energy to moles of octane using the conver- sion factor found in step 2, and then convert from moles of octane to mass of octane using the molar mass. GIVEN: 1.0 x 10¹¹ kJ FIND: kg C8H18 CONCEPTUAL PLAN (1) Write the balanced equation. (2) (3) AHx AHi's AHxn npAH (products) - Σn,AH; (reactants) kJ mol C8H18 114.22 g C8H18 mol CH18 Conversion factor to be determined from steps 1 and 2 RELATIONSHIPS USED molar mass C8H18 = 114.22 g/mol 1 kg = 1000 g g C8H18 1 kg 1000 g kg C8H18
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
1 mol C8H18 50741 kJ ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Let the two primes p= 41 and q = 17 be given as set-up parameters for RSA. 1. Which of the parameters e= 32,e2 = 49 is a valid RSA exponent? Justify your choice. 2. Compute the corresponding private...
-
A psychologist conducts an experiment on rats. She puts a rat in a cage having three rooms labeled 1, 2, and 3, as shown in Figure below. L Figure: a cage with 3 rooms and 5 doors. The rats are...
-
Melissa Ostwerk, the new controller of TurboDrives, Inc., has just returned from a seminar on the choice of the activity level in the predetermined overhead rate. Even though the subject did not...
-
Darlene Cook Company engaged in the following transactions during the month of July: July 1 Acquired land for $10,000. The company paid cash. 8 Billed customers for $3,000. This represents an...
-
Which of the following accounts is not closed? a. Salary Expense b. Service Revenue C. Accumulated Depreciation, Equipment d. Dividends
-
Suppose GNC, a vitamin and supplement supplier, would like to investigate the relationship between the size of an order and the age of the customer who ordered it. This information could allow GNC to...
-
Question 1 (of 2): MicroTech Limited Case Study MicroTech Limited (MT)' is a leading manufacturer of computers, notebooks and printers. MT's strength lies in research and development and, is...
-
One way to evaluate fuels with respect to global warming is to determine how much heat they release during combustion relative to how much CO 2 they produce. The greater the heat relative to the...
-
Which process is endothermic? a) The evaporation of water from the skin b) The burning of candle wax c) The oxidation of iron in a chemical hand warmer d) The combustion of natural gas in a stove
-
Consider the All-Green energy company in Problem 5, Section 9.2. All Green has considered asking a marketing research group to perform a market research study. Within one month, this group can report...
-
A 26-year-old man presents to the Accident and Emergency Unit with groin pain that has come on over the past few hours, which is severe in intensity and leaves him unable to walk. He denies dysuria,...
-
A 26-year-old who is taking long-term oral prednisolone (10 mg/day) therapy comes to the Emergency Department because he is worried, as his brother has been admitted to the hospital with acute...
-
Product Omega has revenue of $ 1 9 4 , 5 0 0 , variable cost of goods sold of $ 1 1 5 , 7 0 0 , variable selling expenses of $ 3 2 , 5 0 0 , and fixed costs of $ 6 1 , 8 0 0 , creating an operating...
-
A corporation is trying to determine if it is exempt from the personal holding company tax. Which code section and subsection indicates what types of entities are not considered personal holding...
-
A 22-year-old man presents to the Emergency Department with a painless ulcer affecting his glans penis. He admits to unprotected sexual intercourse with a man some two weeks earlier. There is no...
-
A few years ago the U.S. dollar equivalent of a foreign currency was $1.2167. Today, the U.S. dollar equivalent of a foreign currency is $1.3310. Determine the percentage change of the euro between...
-
The trade-off theory relies on the threat of financial distress. But why should a public corporation ever have to land in financial distress? According to the theory, the firm should operate at the...
-
The A-36 steel pipe is subjected to the axial loading of 60 kN. Determine the change in volume of the material after the load is applied. 30 mm 40 mm 60 kIN 60 kN 0.5 m
-
Air is pumped into the steel thin-walled pressure vessel at C. If the ends of the vessel are closed using two pistons connected by a rod AB, determine the increase in the diameter of the pressure...
-
Determine the increase in the diameter of the pressure vessel in Prob. 1053 if the pistons are replaced by walls connected to the ends of the vessel. Problem: 10-53 Air is pumped into the steel...
-
30) The function for a current is I(t) = 9 cos(120t) where / is the current, in amperes, and t is the time in seconds. a) Find the current when t = 6 s. b) The electromotive force, E(t), in a...
-
QUESTION 13. [7pts] Consider f(x) = 3 - 2e=-=-1. Answer and show the work for the below questions, and use them to graph f. a) Find the domain of f. b) Find the assymptotes (vertical and horizontal),...
-
A company has determined their yearly cost function, in dollars, to be given by C(x) = 2,200x5/2 6x2+5x-4 when x pounds of product are made. Find the marginal cost (in dollars per pound) when 5...

Study smarter with the SolutionInn App


